With an innovative drug delivery mechanism that aims to help save millions of lives by improving the onset of action or reducing or eliminating side effects of approved therapeutics, Aspire Biopharma (NASDAQ: ASBP) is shaping the future of medicine.


Download The Corporate Presentation
Overview
Aspire Biopharma (NASDAQ: ASBP) engages in the business of developing and marketing novel disruptive technology for the sublingual delivery of drugs and other products.
- Patent-pending drug delivery technology for rapid sublingual absorption and entry into the bloodstream of drugs and supplements
- Enhances the pharmacokinetic performance of Active Pharmaceutical Ingredients (“APIs”), supplements and nutraceuticals into the bloodstream, increasing bioavailability and improving speed of onset
- Multiple ℞ applications in heart attack/stroke, anxiety, nausea, and others
- Can be applied in multiple oral/intraoral product formats such as tablets, capsules, oral suspensions, and others
- Contract Manufacturing Relationships
- Lead Rx Product: Sublingual High Dose Aspirin
- Lead Consumer Product: BUZZ BOMB™ caffeine
- Focused on commercialization through partnerships, licensing and internal development
Corporate & Financial Milestones in 2026
- Share Repurchase Authorized the repurchase of up to $5.0 million of the Company’s common stock.
- Capital Injection: The Company secured $21 million in private placement financing in its February 2026 and April 2026 preferred stock offerings.
- Exchange Compliance: Aspire regained compliance with Nasdaq's minimum stockholders' equity requirement in April 2026 and has received confirmation from Nasdaq.
- Transformative Acquisition: On April 15, 2026, Aspire entered a Binding Letter of Intent to acquire DCS, a global automotive supplier, for approximately $30 million in cash.
- Commitment Letter: The Company secured a Commitment Letter from a leading financial institution of up to $22.5M to finance the proposed acquisition of DCS.
Aspire Biopharma Clinical Pipeline
- Sublingual Aspirin: Aspire's lead candidate, a fast-acting high-dose sublingual aspirin for acute heart attacks, showed it could inhibit platelet aggregation in under two minutes—4 to 5 times faster than chewed tablets.
- FDA Milestones: The Company plans to file a New Drug Application (NDA) via the 505(b)(2) pathway near the end of 2026 for its lead Aspirin candidate.
- Expanded Pipeline: Formulation and development underway for generic sublingual versions of anti-anxiety drug (Xanax®), anti-nausea drug (Zofran®), anti-platelet drug (Plavix®), as well as others
- Intellectual Property: The Company now has Patents filed and pending on eleven different drugs and supplements, including the filing of an omnibus patent application with the U.S. Patent and Trademark Office for its sublingual delivery technology for several key drug classes.
Buzz Bomb Subsidiary Expansion
- Product Launch: BUZZ BOMB™ officially launched its 50mg sublingual caffeine stick packs in four flavors (Tropical Fruit, Mixed Berry, Peach Mango, and Coffee Mocha).
- Buzz Bomb Growth: Launched a new convenience store pack, rebranded with "science-forward" packaging.
- Marketing Momentum: Brand ambassador Ashley Paulson achieved a world-record performance at the recent 100-mile Jackpot Ultra Running Festival, driving increased sales and brand awareness.
- Retail Partnerships:
o National: Partnered with TruLife Distribution to manage and coordinate the sales, distribution and merchandising of BUZZ BOMB™ to retailers nationwide.
o Regional: Signed agreements with Interwest Brokerage to scale BUZZ BOMB™'s retail footprint across the grocery, convenience, and alternate channel sectors throughout the Intermountain West.
Drug and Supplement Delivery Technology Innovator
1. Fast Acting
Powder-form medication developed using our patent-pending technology enters the bloodstream in a fraction of the time as compared to oral tablets and capsules
2. Easy to Use
Dissolves easily under the tongue
3. Dosage Management
Drugs do not first pass through the liver and are not metabolized like swallowed products
4. Bypasses the Digestive Tract
Reduces or eliminates adverse reactions in the gastrointestinal tract
5. Replaces pills and tablets
Easy for patient or caregiver to administer
Aspire Patent and Patent Pending Portfolio
Sublingual Aspirin Product Candidate
- Filed provisional patent applications with the USPTO on March 31, 2023 and October 2, 2024, respectively, pertaining to Oral Mucosal Formulations of Aspirin that utilize Aspire’s fast acting formulation.
Omnibus Patent Filing
- Filed an omnibus patent application with the U.S. Patent and Trademark Office (“USPTO”) for its sublingual delivery technology October 2025.
- Application seeks patent protection of Aspire’s proprietary technology in a variety of classes of drugs and other substances focusing exclusively on protecting the Company’s advanced sublingual drug delivery system,
- An omnibus claim is a statement in a patent application that describes the unique features of an invention and defines the scope of protection for the patent. It is a formal way of summarizing the points of novelty of the invention and is usually followed by a fixed form. The omnibus patent can provide IP protection in new areas/classes in addition to the patents already filed by Aspire.
Non-Aspirin Drug Product Candidate Patents Filed January 2026
▪ Alprazolam (Generic Xanax®): Formulation for sublingual administration of drug for treatment of anxiety. Provisional patent filed January 2026.
▪ Clopidogrel (Generic Plavix®): Antiplatelet to prevent blood clots. Provisional patent filed January 2026.
▪ Odansetron (Generic Zofran®): Antiemetic medication that prevents nausea and vomiting from chemotherapy, radiation, and surgery. Provisional patent filed January 2026.
▪ Meclizine: Antihistamine used primarily to prevent and treat motion sickness (nausea, vomiting, dizziness) and vertigo. Provisional patent filed February 2026.
Aspire’s Sublingual Aspirin Product Candidate
ASBP’s sSublingual aAspirin pProduct addresses cardiology emergencies and is intended for the treatment of suspected acute myocardial infarction (AMI, blockage of blood flow to heart muscle causing damage or death of heart tissue – commonly known as a “heart attack”). There are an estimated 18 million Americans living with coronary artery disease with approximately 800,000 per year experiencing an AMI leading to 300,000 deaths.
Sublingual Aspirin Product Study Design and Top-Line Results
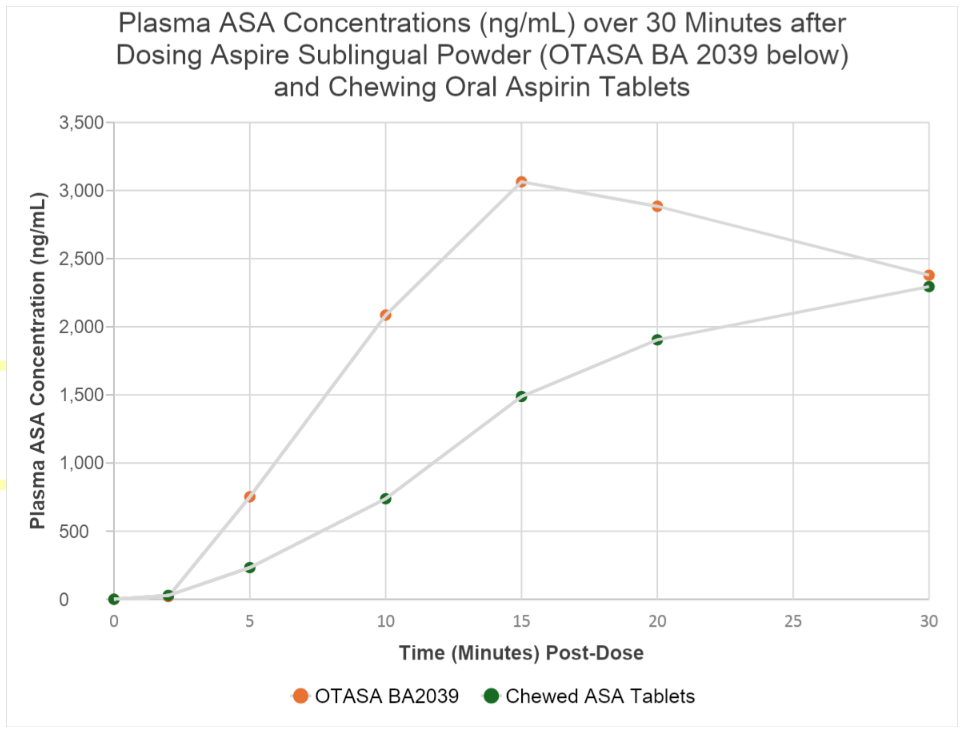
▪ The primary objective of the clinical trial was to evaluate the bioavailability of acetylsalicylic acid (ASA, the active antiplatelet form of aspirin) in plasma over eight hours after dosing.
▪ Randomized, crossover bioavailability trial to assess the safety, tolerability, pharmacokinetics (PK) and pharmacodynamics (PD) of Aspire’s investigational new powder formulation sublingual aspirin product compared to chewed uncoated aspirin tablets in healthy adults.
▪ Aspire’s sublingual aspirin product produced higher and more rapid mean plasma concentrations of ASA, compared to chewed aspirin tablet.
Significant improvement in absorption was evident within five minutes and continued throughout the first half-hour after dosing.
Achieved therapeutic doses 2-3 times faster than chewable aspirin tablets
▪ Aspire product was safe and well-tolerated by patients, and no adverse events were reported.
Benefits of “rapid absorption” aspirin
▪ Address and limit heart attack and stroke
▪ Allow high dose absorption for pain management including quick headache relief, post surgery, cancer pain management, and general pain relief
Additional Important Clinical Trial Findings
▪ Aspire's sublingual aspirin formulation significantly inhibited serum thromboxane B2 (TxB2) within the first two minutes after dosing
▪ TxB2 is a biomarker indicating aspirin's effect on platelet aggregation, the clumping of platelets that leads to dangerous blood clots; average person has concentration at 120 ng/mL.
▪ This rapid inhibition is critical during suspected heart attacks, when clots blocking heart arteries can cause permanent muscle damage
▪ Lab results showed Aspire's product acted more than five times as fast as the current recommendation for chewed aspirin tablet treatment
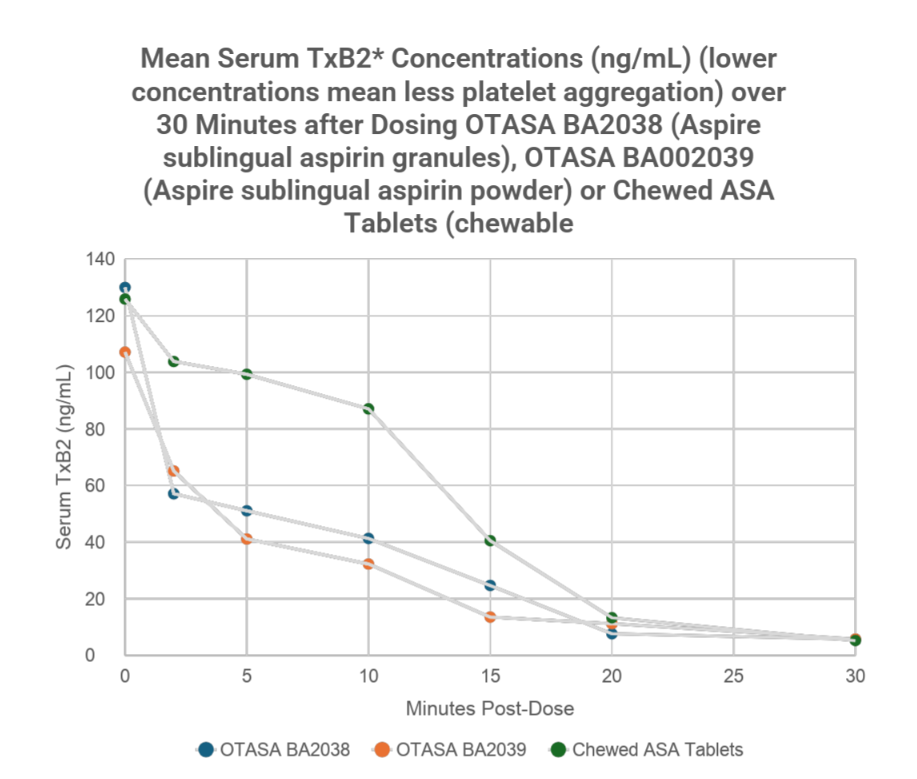
* TxB2 is a stable metabolite of thromboxane A2 (TxA2), a potent chemical that promotes platelet clumping and vasoconstriction. Measuring low levels of TxB2 confirms that aspirin is working as intended
In the planned initial launch of its sublingual products, Aspire has focused on the delivery of aspirin, which may be the most studied and accepted analgesic and anti-inflammatory.
Aspirin is over a century old and is traditionally available in several forms, including effervescence, powder, capsule, and tablet. Over 100 years of documented safety and efficacy data is readily available.
The Company plans to seek FDA 505(b)(2) Fast Track designation for the prescription strength sublingual aspirin. The 505(b)(2) pathway specifically benefits new drugs that are similar to already approved drugs but have slight variations in formulation or administration routes. Aspire can reference the safety and efficacy data of the original innovator drugs–which are already FDA-approved–accelerating the approval process and reducing associated costs.
Sublingual Aspirin Product Commercialization and Go-to-Market Strategy
1 Launch product initially in the Rx market, likely through licensing or partnering agreements
2 Partner with an experienced end-to-end marketing and distribution firms
3 Significant potential licensing opportunities with leading drug manufacturers
The Market Opportunity
Global Analgesics Market
Valued at $47.32B in 2023 and projected to reach $75.73B by 2032, growing at a compound annual growth rate (CAGR) of 5.39% over the forecast period 2024-2032. (Source: SNS Inside

BUZZ BOMB™ Caffeine Products
Unlike traditional energy drinks or pills, BUZZ BOMB™ is a new and exciting caffeine product delivered in a single-serving stick pack of dry powder sprinkled under the tongue. This method provides flavored caffeine quickly without the hassle of mixing with water or consuming typical caffeine sources like energy drinks, coffee, or soda.
BUZZ BOMB™ features 50mg of caffeine and is currently offered in four delicious flavors: Tropical Fruit, Mixed Berry, Peach Mango, and Coffee Mocha. Designed for athletes, professionals, and the everyday person needing a rapid boost, BUZZ BOMB™ provides a precise serving of caffeine in easy-to-use single serving stick packs.
- Nano technology delivers caffeine rapidly to the blood stream, bringing its unique disruptive benefits to the caffeine market
- Initial offering of four flavor options
- Marketing plan focused on cost-effective multi-channel digital strategy targeting primary influencers, direct response sales and traditional retail sales channels launched
- Global pre-workout supplements market size is expected to reach $27.97 billion by 2030, registering a CAGR of 5.9% from 2025 to 2030
BUZZ BOMB™ Disruptive Characteristics
- Speed - works nearly immediately (less than 2 minutes) vs. 20-30 minutes
- Convenience – small, easy to use single-use packets (no mixing and measuring for beverages)
- Energy management - use as needed to precisely manage caffeine intake (50mg increments)
- Single Safe Active Ingredient – well-known benefits and use of caffeine
- Low manufacturing & packaging costs - competitive pricing with high margin potential
Global Pre-Workout Market

Supplements market valued at $19.90B in 2023. Estimated to reach $29.77B by 2032, growing at a CAGR of 4.58% during the forecast period (2024–2032). (Source: Straits Research)
- Rising fitness culture and gym memberships
- Consumer focus on performance and recover
- Expanding demographics (women, Gen Z, casual exercisers)
- Growth of RTD ("Ready To Drink"- no mixing) formats & natural formulations
- Digital fitness and influencer-led marketing
Management
Kraig Higginson
Chief Executive Officer
Mr. Higginson brings to Aspire BioPharma, Inc. extensive experience in early-stage growth company management as well as vast public market expertise. Mr. Higginson founded and served as Chief Executive Officer of VIA Motors, Inc., a hybrid electric vehicle company from November 2010 to January 2015, which achieved more than a $1 billion market cap. From October 2003 until November 2010, he founded and served as Chairman of the Board of Directors of Raser Technologies, a NYSE company which achieved a $1.7 billion market cap. Earlier Mr. Higginson also founded American Telemedia Network, Inc., a publicly traded NASDAQ company that developed a nationwide satellite network broadcasting data for large corporations, as well as video programming and advertising to shopping centers and malls. He served as President and Chief Executive Officer of American Telemedia Network from 1984 through 1988. Mr. Higginson has been a leading entrepreneur for several decades.
Ernest J. Scheidemann, Jr.
CPA, MBA - Chief Financial Officer
Mr. Scheidemann brings over 20 years of CFO experience leading financial and operational teams of both publicly traded and private companies, in Fortune 500, mid-cap, mega start-ups and fast-paced entrepreneurial organizations in a variety of industries, and has served on boards of health care services related non-profits. Mr. Scheidemann’s expertise includes building and leading highly skilled accounting and finance teams, debt and equity financing, GAAP accounting, strategic planning, M&A, profitability maximization, financial processes, and SEC reporting. Previously, Mr. Scheidemann was the Chief Financial Officer for Benchmark Builders, Inc., ASG Technologies, Inc., WCI Communities, Inc., and Walker Digital, LLC, and earlier in his career served as also the Controller for AT&T’s $23 billion Consumer Services division. Mr. Scheidemann is a licensed Certified Public Accountant (CPA) in the State of New Jersey and holds a Certified Global Management Accountant designation (CMGA) and a Certification in Financial Forensics (CFF), each from the American Institute of CPAs.
In Summary…
Aspire Bio Labs (NASDAQ: ASBP)
- Multiple ℞ applications in heart attack/stroke, anxiety, nausea, antiplatelet drug to prevent blood clots, and others
- Large Addressable ℞ Markets
- High Dose Aspirin Prescription Product: Trial demonstrated dramatically higher and more rapid therapeutic impact compared to standard chewed aspirin tablets, safe and well-tolerated. Aspire's sublingual aspirin formulation significantly inhibited serum thromboxane B2 (TxB2) within the first two minutes after dosing. This rapid inhibition is critical during suspected heart attacks, when clots blocking heart arteries can cause permanent muscle damage.
- Expect to file in 2026 for FDA 505 (b)(2) Fast Track drug approval process for high dose aspirin
- BUZZ BOMB™, New Pre-Workout Supplement Launched in Q3 2025: Commercial revenue in 2026
- Global pre-workout supplements market size: Expected to reach $27.97 billion by 2030
- Focused on drug product commercialization through partnerships and licensing
- Proprietary Patent-Pending Technology: Enables ability to pursue broadened applications, using our solubility process, which can be used with other drug and supplement compounds
Aspire Biopharma (NASDAQ: ASBP) has developed a disruptive technology for delivering drugs and other products sublingually with the goal of bringing multiple already approved therapeutics to the market with improved onset of action or a reduction in side effects. While pursuing drug candidates in large markets, Aspire expects to grow its revenue in 2026 from the commercial launch of BUZZ BOMB™ it’s caffeine supplement. Now may be the ideal time to have the company on your watch list!

Learn More about Aspire Bio Labs. by gaining access to the latest corporate presentation
Download PRESENTATIONTHIS IS A PAID ADVERTISEMENT
NO INVESTMENT ADVICE
Copyright 2025 © SCDalerts.com is owned and operated by the owner of SCD Media LLC.
Disclaimer and Privacy For more Information please contact info@smallcapsdaily.co
This website provides information about the stock market and other investments. This website does not provide investment advice and should not be used as a replacement for investment advice from a qualified professional. This website is for informational purposes only. The Author of this website is not a registered investment advisor and does not offer investment advice. You, the reader, bear responsibility for your own investment decisions and should seek the advice of a qualified securities professional before making any investment. Nothing on this website should be considered personalized financial advice. Any investments recommended here in should be made only after consulting with your personal investment advisor and only after performing your own research and due diligence, including reviewing the prospectus or financial statements of the issuer of any security.
SCD Media, its managers, its employees, affiliates, and assigns (collectively "The Company") do not make any guarantee or warranty about the advice provided on this website or what is otherwise advertised above. To the maximum extent permitted by law, the Company disclaims all liability in the event any information, commentary, analysis, opinions, advice and/or recommendations provided herein prove to be inaccurate, incomplete, or unreliable, or result in any investment or other losses.
You received this message as part of your subscription to SCD Alerts.
SCD Alerts is a financial news and information website. We do not directly sell any products or offer any personal financial advice, nor do we advocate the purchase or sale of any security or investment for any specific individual. We also do not make any guarantee or warranty about what is advertised above.
If you have questions or concerns about a product you’ve seen in one of our emails, we encourage you to reach out to that company directly. Disclaimer – Always do your own research and consult with a licensed investment professional before investing. This communication is never to be used as the basis of making investment decisions and is for entertainment purposes only. At most, this communication should serve only as a starting point to do your own research and consult with a licensed professional regarding the companies profiled and discussed. Conduct your own research. This newsletter is a paid advertisement, not a recommendation nor an offer to buy or sell securities. This newsletter is owned, operated, and edited by SCD Media. Any wording found in this e-mail or disclaimer referencing “I” or “we” or “our” or “SCD” refers to SCD Media. Our business model is to be financially compensated to market and promote small public companies. By reading our newsletter and our website you agree to the terms of our disclaimer, which are subject to change at any time. We are not registered or licensed in any jurisdiction whatsoever to provide investing advice or anything of an advisory or consultancy nature and are therefore unqualified to give investment recommendations. Companies with low prices per share are speculative and carry a high degree of risk, so only invest what you can afford to lose. By using our service, you agree not to hold our site, its editor’s, owners, or staff liable for any damages, financial or otherwise, that may occur due to any action you may take based on the information contained within our newsletters or on our website. We do not advise any reader to take any specific action. Losses can be larger than expected if the company experiences any problems with liquidity or wide spreads. Our website and newsletter are for entertainment purposes only.
Never invest purely based on our alerts. Gains mentioned in our newsletter and on our website may be based on end-of-day or intraday data. This publication and its owners and affiliates may hold positions in the securities mentioned in our alerts, which we may sell at any time without notice to our subscribers, which may have a negative impact on share prices. If we own any shares, we will list the information relevant to the stock and the number of shares here.
We do not own any shares in ASBP. We have been currently compensated up to Twenty Five Hundred Dollars Cash ($2,500) via bank wire transfer from a third-party Interactive Offers, LLC for landing page hosting for ASBP with a start date of 3/10/2025. SCD’s business model is to receive financial compensation to promote public companies. This compensation is a major conflict of interest in our ability to be unbiased regarding our alerts. Therefore, this communication should be viewed as a commercial advertisement only. We have not investigated the background of the hiring third party or parties. The third party, profiled company, or their affiliates likely wish to liquidate shares of the profiled company at or near the time you receive this communication, which has the potential to hurt share prices. Any non- compensated alerts are purely for the purpose of expanding our database for the benefit of our future financially compensated investor relations efforts.
Frequently companies profiled in our alerts may experience a large increase in volume and share price during investor relations marketing, which may end as soon as the investor relations marketing ceases. The investor relations marketing may be as brief as one day, after which a large decrease in volume and share price is likely to occur. Our emails may contain forward looking statements, which are not guaranteed to materialize due to a variety of factors. We do not guarantee the timeliness, accuracy, or completeness of the information on our site or in our newsletters. The information in our email newsletters and on our website is believed to be accurate and correct but has not been independently verified and is not guaranteed to be correct. The information is collected from public sources, such as the profiled company’s website and press releases, but is not researched or verified in any way whatsoever to ensure the publicly available information is correct.
Furthermore, SCD often employs independent contractor writers who may make errors when researching information and preparing these communications regarding profiled companies. Independent writers’ works are double-checked and verified before publication, but it is certainly possible for errors or omissions to take place during editing of independent contractor writer’s communications regarding the profiled company(s). You should assume all information in all of our communications is incorrect until you personally verify the information, and again are encouraged to never invest based on the information contained in our written communications.
The information in our disclaimers is subject to change at any time without notice.
