Developing OTC products focused on hair, skin, and sexual health, this rising NASDAQ company recently posted its strongest revenue numbers to date…

Jupiter Wellness, Inc. (NASDAQ: JUPW) ) is innovating health science into revolutionary products aimed at skin, hair, sexual wellness, and general health.

Download Research Report

Clinical Pipeline
Jupiter Wellness, Inc. (NASDAQ: JUPW) is proving to be an exciting player to watch in the bustling medical wellness industry as it has many exciting acquisitions!
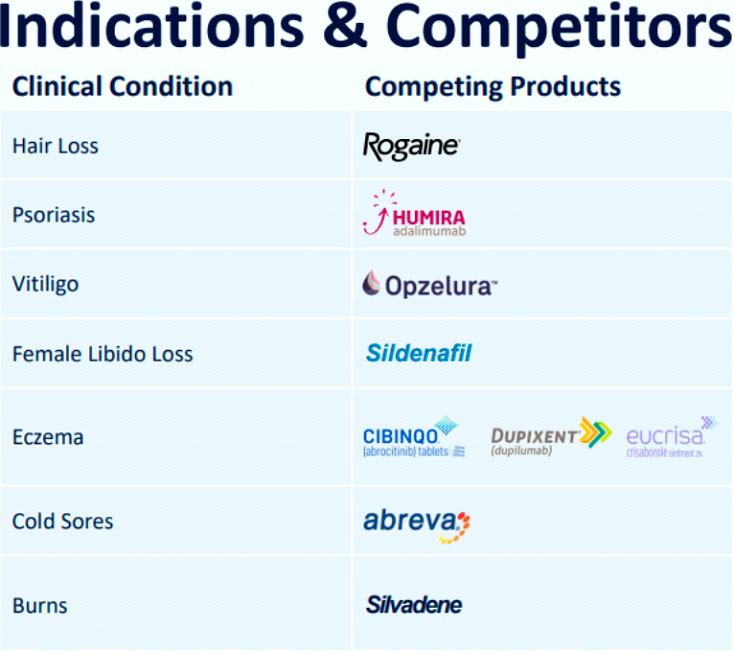
JUPW is targeting tremendous markets with well-known rivals that include Johnson & Johnson owned Rogaine, Abbott Laboratories owned Humira, and Pfizer’s Sildenafil.
Zooming in on the Women’s Sexual Wellness Category with RJ-101!

Female audiences have been routinely placed in the shadows by major CPG companies when it comes to addressing and serving their core sexual wellness needs.
JUPW is taking notice of this key and very important demographic that represents massive spending power.
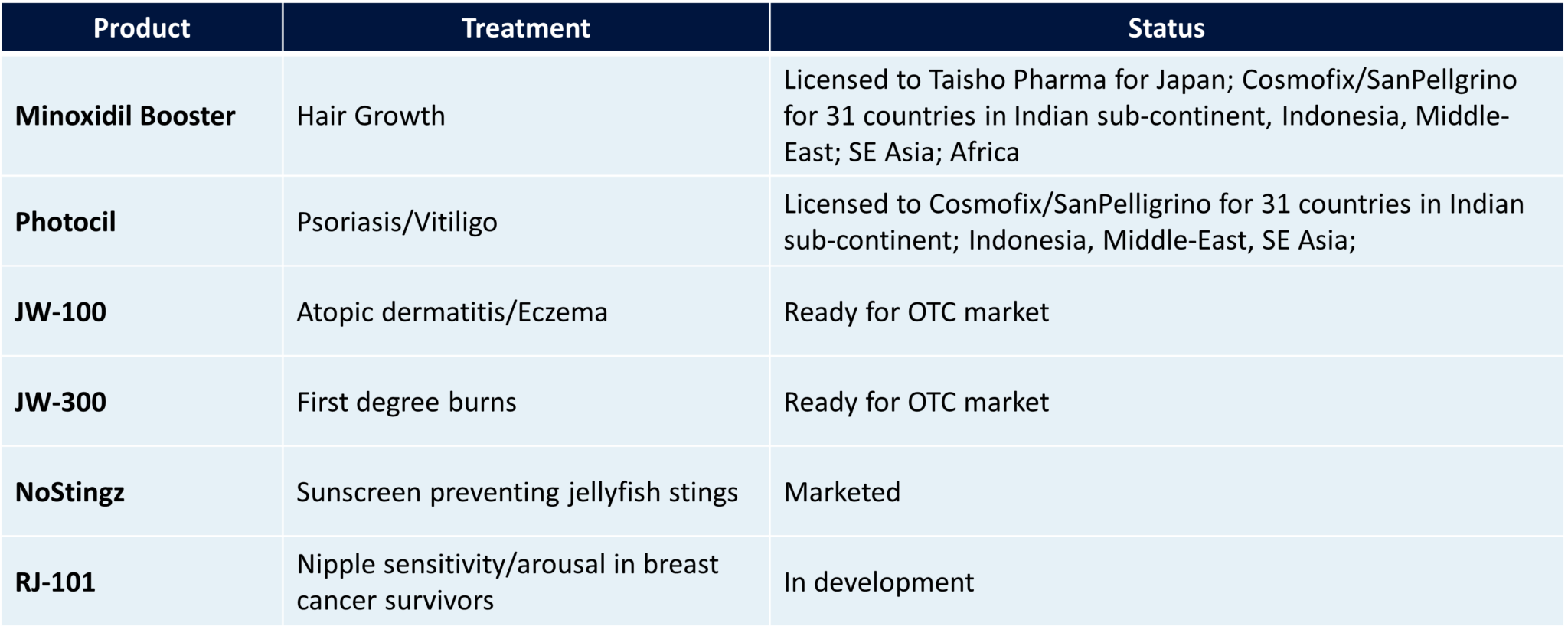
One of the most exciting developments the company has is RJ-101 – which could address a wide range of clinical indications including: female orgasmic disorder, nipple neuropathies after breast augmentation, and orgasmic adverse events in breast cancer survivors.
It is covered by two issued U.S. patents - patent number 10,357,452 and patent number 10,064,816.
“We are excited to complete the development of RJ-101 started by Dr. Andy Goren and the team at Applied Biology and to bring this novel approach to the market. I believe this product will be an excellent treatment for people with breast cancer.”
RJ-101 is currently in pre-revenue.
The Company plans to initially develop this product for the treatment of nipple neuropathies in breast cancer patients through the filing of an IND.
"This strategic asset purchase agreement adds immediate revenues to Jupiter in Q2 2022 and contributes positively towards EBITDA with no upfront cash or stock acquisition cost to our company. The acquired contracts and related assets enhance Jupiter's position as a research and development powerhouse while allowing us to enter the lucrative clinical research organization business."
- Jupiter Wellness CEO, Brian John
As a result of the pandemic, many women have made significant lifestyle resets at both an individual and collective level around the world.
Some of these changes have included how women are seeking, experiencing and demanding better sex in their day-to-day lives. Women are now giving themselves permission to speak freely about their sexual wellness needs and desires.
The US sexual wellness market is expected to grow at a CAGR of 8.27% during the period 2020−2026. According to Business Wire, the market is predicted to reach USD $125.1 billion by 2026!
Beauty Independent said it best, “Consumers can buy lube at Nordstrom, vibrators at Holt Renfrew and arousal oils at Urban Outfitters rather than having to patronize sex shops for intimate goods. And the sales of sex products show no signs of slowing down.”
Tackling the Enormous Skincare Market
Skincare, as a market segment, is growing faster than any other part of the beauty industry.
Just think of what our skin can go through, from bug bites to sun burns, or even more serious conditions such as acne, psoriasis, and eczema.
Skin diseases currently make up 1.79% of the global disease burden.
The American Academy of Dermatology (AAD) reports that 84.5 million Americans — 1 in 4 — are impacted by skin diseases.
According to Precedence Research, the global skin care products market size is predicted to surpass around US$ 204.62 billion by 2030 and growing at CAGR of 4.6% from 2021 to 2030.
Skincare has become an increasingly popular trend as more and more people are looking for greener, healthier products that address specific skincare needs.
JUPW is quickly becoming a leader in the medical wellness scene with an impressive and diverse development pipeline that includes products to address psoriasis, eczema, burns, herpes, and cold sores.
In recent, exciting news, JUPW announced the acquisition of certain assets and IP of Applied Biology, Inc. a leading biotechnology company specializing in hair and skin science.
Beyond this, the acquisition also expands JUPW’s pipeline by including treatments for:
- Alopecia - pipeline for the treatment of traction alopecia, androgenetic alopecia, and chemotherapy-induced alopecia.
- Psoriasis - A topical treatment for psoriasis to be launched in late 2022.
- Women's Health - A novel treatment for female sexual dysfunction in the clinical stage was recently granted 2 patents.
JUPW also recently announced it has concluded an Asset Purchase Agreement with Ascent Clinical Research, Inc. through which it has acquired certain clinical assets including clinical trial research contracts worth $3 million in annual revenues for a purchase price of 5% royalties on future net revenues generated by the assets.
The company has signed an agreement with India-based companies Cosmofix Technology Pvt Ltd and Sanpellegrino Cosmetics Pvt Ltd for the manufacture, sale, and distribution of three proprietary products related to its recent acquisition of Ascent Clinical Research.
This is huge news as JUPW is entitled to royalties of between 10-12% on revenues for the three products with guaranteed annual minimum royalty payments for the duration of the agreement in 31 countries in Asia and the Middle East including India, the Philippines, Malaysia, Kuwait, Morocco, and Saudi Arabia!

Eczema (JW-100): Topical treatment for atopic dermatitis (eczema).
According to Jupiter Wellness, studies showed that JW-100 cleared or reduced eczema symptoms after two weeks of use. Results suggest that JW-100 may potentially prove superior to existing prescription drugs.
A Phase 3, double-blind, placebo-controlled multicenter trial is underway to evaluate the superiority of JW-100 to Eucrisa a Food and Drug Administration (FDA)-approved topical drug) in mild to moderate eczema.
Psoriasis and vitiligo (Photocil): Topical treatment for psoriasis and vitiligo.
JUPW says Photocil safely and effectively permits phototherapy treatments at home by blocking harmful radiation and allowing the passage of therapeutic ultraviolet (UV) radiation.
It is anticipated that Photocil will be available online in the U.S. in the fourth quarter of 2022 as an FDA-approved over-the-counter product under a United States Pharmacopeia (USP) monograph for psoriasis and vitiligo.
The company reports that clinical trial results showed that Photocil cream acts as a barrier to nontherapeutic radiation from the sun when skin is exposed to direct daylight.
Burns (JW-300): Topical treatment of first-degree burns and sun exposure.
JUPW reports that JW-300 was shown to significantly lower the incidence of burns in patients exposed to UV radiation.
JW-300 is being evaluated for sale as an after-sun consumer product to complement the company’s NoStingz sunscreen — topical protection from jellyfish, sea lice, and ultraviolet A (UVA)/ultraviolet B (UVB) rays — and legacy sun care product lines.
Cold sores (JW-400): Topical treatment for herpes labialis (cold sores).
A phase 1, a double-blinded placebo-controlled investigational study is being planned.
JUPW has signed a license agreement with Shark Defense Technologies for the exclusive worldwide intellectual property rights to formulas and production methods for jellyfish protective compounds including 4 patents pending!
The agreement includes future collaboration to develop and refine additional formulas.
According to Fortune Business Insights, the global sun care market was worth $13 billion in 2019 and is projected to reach $17 billion by 2027.

In its report, Jellyfish Gone Wild!, the U.S. National Science Foundation estimates that 150 million people are stung by jellyfish each year, with about 200,000 these occurring in Florida, and many scientists believe the number of jellyfish is increasing.
Jellyfish stings can vary in severity from immediate pain, swelling, itching, and tentacle print marks, to more systemic reactions such as difficulty breathing and heart problems that may be life threatening, according to the Mayo Clinic.
JUPW’s NoStingz line includes sprays and lotions for children and adults with SPFs ranging from 30 to 50, with more SKUs to be added in the near future. Each product’s dermatologically tested, hypoallergenic, scientifically proven, water-resistant formula prevents stings from most jellyfish and sea lice, gives sunray and UV protection, is non-toxic for the marine environment, and is packaged in a recyclable bottle.
NoStingz is just as good, if not better than the major name brands on the market but with the added benefit of protecting users from jellyfish and sea lice stings.
“If you have a store near a beach, your customers want this product. If you visit the beach with your kids, you need this product."


Hair Loss (Minoxidil Booster) is a Topical treatment designed to improve Minoxidil efficacy.
JUPW reports minoxidil is the only FDA-approved topical drug for treating common hair loss (androgenetic alopecia)!

The company says clinical studies of Minoxidil Booster have shown it increases the enzymes needed for minoxidil to work, sulfotransferase enzymes, by up to seven times over a two-week period.
Minoxidil Booster has been licensed to Taisho Pharmaceutical Holdings, a $2.6 billion company and Japan’s leading seller of minoxidil products. It expects to launch the product commercially in 2023.
It has also been licensed to India-based Cosmofix Technovation Pvt Ltd and Sanpellegrino Cosmetics and is expected to be launched soon.
Revenues are Skyrocketing
JUPW generated $3,000,582 in revenues for the three months ended June 30, 2022 compared to $595,088 revenues in the three months ended June 30, 2021.
The company is now experiencing a greater demand for their products…
Jupiter Wellness is on track with its 2022 roadmap and finished the first half of 2022 with momentum and our strongest revenue numbers to date.
Go to Market Plan
Go-To-Market Strategy: Focus on B2B and B2C sales/distribution by following channels:
- MMR: Sales team to continue developing and maintaining relationships with mass merchandise retailers like Walmart, Target, CVS, Walgreens, etc.
- Wholesale: Expand contracted brokers/reps to saturate independent retailers like pharmacies, doctor offices, drug stores, etc.
- E-Comm: Execute shrewd integrated marketing campaigns with a heavy focus on brand awareness to increase website conversions and build customer lifetime value.
- Strategic IP: Focus on licensing formulations worldwide.
Growth Plan for New Products
- Expand B2B store count and existing shelf space
- Maintain retailer relationships/shelf space as regulation develops
- Increase B2C site visitors with targeted brand awareness campaigns
- Optimize product offering by sales channel
- Focus product innovation on “claims-based” products
- Leverage flagship brand equity with line extensions and cross-merchandising
- Pursue new markets that leverage brands and infrastructure (Licensing opportunities)
- Optimize cost structure to increase margins and EBITDA
- Invest in science/research to support product claims
- Build and maintain relationships with regulators such as the FDA
THE BOTTOM LINE

JUPW’s clinical pipeline is competing against some of the biggest names in skin care, hair loss, and sexual wellness.
As an emerging company on the NASDAQ, their story seems to be only now unfolding with product launches geared up for the new future…
Revenues are climbing… demand for products is growing…. Zooming in on big distributors such as Target, Walgreens, and CVS could quickly make the company into a household name across multiple markets.
Jupiter Wellness, Inc. (NASDAQ: JUPW) has an innovative and exciting pipeline in several multi-billion-dollar markets that make this wellness company very exciting to watch as it reaches new heights!

THIS IS A PAID ADVERTISEMENT
NO INVESTMENT ADVICE
This website is wholly owned by scd media llc (d/b/a “smallcapsdaily.com”). Our reports are advertorials and are for general information purposes only. Never invest in any stock featured on our site or emails unless you can afford to lose your entire investment. This disclaimer is to be read and fully understood before using our services, joining our email list, as well as any social networking platforms we may use. Please note as well: Small Caps Daily and its employees are not Registered Investment Advisors, broker-dealers, or member(s) of any association for other research providers in any jurisdiction whatsoever. release of liability: through use of this website, viewing or using you agree to hold Small Caps Daily, its operators, owners, and employees harmless and to completely release them from any and all liability due to any and all loss (monetary or otherwise), damage (monetary or otherwise), or injury (monetary or otherwise) that you may incur. The information contained herein is based on sources that we believe to be reliable but is not guaranteed by us as being accurate and does not purport to be a complete statement or summary of the available data. Small Caps Daily encourages readers and investors to supplement the information in these reports with independent research and other professional advice. All information on featured companies is provided by the company profiled or is available from public sources and Small Caps Daily makes no representations, warranties, or guarantees as to the accuracy or completeness of the disclosure by the profiled company. None of the materials or advertisements herein constitute offers or solicitations to purchase or sell securities of the companies profiled herein and any decision to invest in any such company or other financial decisions should not be made based upon the information provided herein. Instead, Small Caps Daily strongly urges you to conduct a complete and independent investigation of the respective companies and consideration of all pertinent risks. Small Caps Daily’s full disclosure is to be read and fully understood before using Small Caps Daily's website, or joining Small Caps Daily's email or text list. From time to time, Small Caps Daily will disseminate information about a company via website, email, sms, and other points of media. By viewing Small Caps Daily's website and/or reading Small Caps Daily's email or text newsletter you are agreeing to this ----> https://smallcapsdaily.com/disclaimer/. All potential percentage gains discussed in any communications are based on calculations from the low to the high of the day. We are engaged in the business of marketing and advertising companies for monetary compensation. In compliance with section 17(b) of the securities act we are disclosing that we have been compensated a fee pursuant to an agreement between scd media llc and sea path advisory. Small Caps Daily was hired for a period beginning july 2022 and ending january 2023 to publicly disseminate information about Jupiter Wellness, Inc. via website, email, and sms. We were paid five thousand usd via ACH. We are also disclosing that Tradigital Marketing Group has been compensated a fee pursuant to an agreement between Tradigital and Jupiter Wellness, Inc. Tradigital was hired for a period beginning july 2022 and ending january 2023 to publicly disseminate information about Jupiter Wellness, Inc., via website, email, and SMS. Tradigital was paid four hundred forty thousand USD via ACH. Tradigital owns three hundred thousand restricted common shares of Jupiter Wellness, Inc., which are eligible for sale on 02/22/2023. For the purpose of this disclaimer, we suggest that you assume we will sell all of our shares once the restriction is lifted on 02/22/2023. Readers are advised to review sec periodic reports: forms 10-q, 10k, form 8-k, insider reports, forms 3, 4, 5 schedule 13d. Small Caps Daily is compliant with the can-spam act of 2003. Small Caps Daily does not offer investment advice or analysis, and Small Caps Daily further urges you to consult your own independent tax, business, financial, and investment advisors. investing in micro-cap, small-cap, and growth securities is highly speculative and carries an extremely high degree of risk. it is possible that an investor's investment may be lost or impaired due to the speculative nature of the companies profiled.the private securities litigation reform act of 1995 provides investors a safe harbor in regard to forward-looking statements. any statements that express or involve discussions with respect to predictions, expectations, beliefs, plans, projections, objectives, goals, assumptions or future events, or performance are not statements of historical fact may be forward-looking statements. forward-looking statements are based on expectations, estimates, and projections at the time the statements are made that involve a number of risks and uncertainties which could cause actual results or events to differ materially from those presently anticipated. forward-looking statements in this action may be identified through the use of words such as projects, foresee, expects, will, anticipates, estimates, believes, understands, or that by statements indicating certain actions & quotes; may, could, or might occur. understand there is no guarantee past performance will be indicative of future results in preparing this publication, Small Caps Daily has relied upon information supplied by its clients, as well as its clients’ publicly available information and press releases which it believes to be reliable; however, such reliability can not be guaranteed. investors should not rely on the information contained on this website. Rather, investors should use the information contained in this website as a starting point for doing additional independent research on the featured companies. the advertisements in this website are believed to be reliable, however, Small Caps Daily and its owners, affiliates, subsidiaries, officers, directors, representatives, and agents disclaim any liability as to the completeness or accuracy of the information contained in any advertisement and for any omissions of material facts from such advertisement. Small Caps Daily is not responsible for any claims made by the companies advertised herein, nor is Small Caps Daily responsible for any other promotional firm, its program, or its structure. Small Caps Daily is not affiliated with any exchange, electronic quotation system, the securities exchange commission, or finra.