This Clinical-stage Biotech Company is Taking Its Revolutionary Platform to Help Eradicate Many Respiratory Viruses: a SINGLE drug treats RSV, FLU/Birdflu, long COVID plus monkey-pox & measles, and may even help lower the risk of metastatic cancer returning in patients following viral infections.
NanoViricides, Inc. (NYSE American: NNVC) is a clinical-stage, global leader in broad-spectrum antiviral nanomedicines is developing therapeutic drugs that are safe and effective, even against variants, using a Revolutionary Platform Technology that does not rely on the host’s immune system. It’s lead drug NV-387 has completed Phase 1a & Phase 1b with no reported adverse effects, indicating excellent safety of Oral Syrup & Oral Gummies.
Broad-Spectrum Antiviral Drug NV-387 Cleared for Phase II Clinical Trial Application by the National Ethics Committee of the Democratic Republic of Congo Read more...
Lead drug, NV-387 addresses a $2.5 Billion to $4.3 Billion market:
- Since it is a potential cure for RSV infection, and since there is no non-toxic drug for general treatment of RSV infection at present; NV-387 meets an unmet need.
- NV-387 is well on its way to becoming the very first drug to treat Measles, showing strong antiviral effectiveness against measles in a humanized animal model study by increasing survival with no signs of toxicity.
- The anti-Influenza activity of NV-387 given orally was substantially superior to all three of the approved anti-influenza drugs (Tamiflu, Rapivab, and Xofluza).
- Bird flu is rampant in poultry in the USA leading to hen culling and soaring egg prices, resulting in a race to find a treatment that works; NV-387 can address it.
- Long COVID also remains a problem for an estimated 17 million adults; an effective antiviral such as NV-387 has already delivered successful Phase 1 results.
- NNVC is initiating a Phase II clinical trial of NV-387 to treat Monkeypox first, because competitors' clinical trials against Monkeypox have failed and NV-387 can potentially be acquired by BARDA for the Strategic National Reserve.

Download The Corporate Presentation

Nanoviricides CEO Dr Anil Diwan joined Steve Darling from Proactive to discuss the company's exploration of using its antiviral candidate, NV-387, for the treatment of MPOX patients under the World Health Organization's (WHO) MEURI protocol.
Watch the video
NV-387 - A novel broad-spectrum antiviral
NanoViricides, Inc. (NYSE American: NNVC)’s lead drug candidate NV-387 (drug product NV-CoV-2), a drug that treats RSV, COVID-19, Long COVID, Influenza, Bird Flu H5N1, and other respiratory viral infections as well as Monkey-pox, has successfully completed Phase 1 clinical trials in healthy subjects with no reported adverse events, even at the highest and repeated dosages. Remarkably, NNVC has been able to develop NV-387 for oral administration already, as well as for injectable and inhalation formulations to enable many modes of use. The Company is currently focused on advancing NV-387 into Phase II human clinical trials for the treatment of RSV infection & monkeypox.
NanoViricides already has a clinical stage broad-spectrum antiviral drug NV-387 that may be effective for treating Measles infection. NV-387 was found to cure lethal RSV infection in an animal model. Measles virus is related to RSV, and both use the heparan sulfate proteoglycan (HSPG) for infection. NV-387 mimics HSPG, presents itself as a decoy to the virus, and is capable of destroying the virus particle thereby stalling the continuous infection and spread of virus in the body. This provides strong rationale for use of NV-387 to combat Measles virus.
Susceptible viruses CANNOT escape NV-387, even as they continue to evolve in the field into variants. Why? Because no matter how much the virus changes, it continues to use the same host-side signature to bind to and cause infection in the hosts, and thus the nanoviricide would be anticipated to continue to be effective even as the virus mutates to generate variants.
Thus NV-387 and other antiviral drugs designed on the nanoviricides platform can be expected to have decades of effective usability against the target viruses similar to the life of current antibiotics against bacterial infections but in stark contrast to current antiviral approaches.
A broad-spectrum antiviral drug such as NV-387 would be a highly desirable drug globally because it would enable treatment by physicians of patients as soon as they present symptoms of a viral disease without waiting for a test to identify a specific type of viral infection. This is reminiscent of how antibiotics are prescribed, without specific infectious agent identification, relying on the ultra-broad-spectrum of the drug.
NV-387’s Extremely Broad Antiviral is a Host Mimetic That Acts As a Decoy To Attract And Trap Many Diverse Viruses, Preventing the Virus from Replication and Reinfection of Other Cells
Over 90% of human pathogenic viruses are known to use one or more "landing sites" that are in the Sulfated Proteoglycans ("SPG") family. A successful host-mimetic nanoviricide drug using SPG as the key feature to attract viruses could theoretically be able to attack most if not all such viruses.
NV-387 is designed to mimic SPG and attack the virus as a cell-mimicking decoy. We have accumulated substantial evidence that in lethal viral infection animal studies, NV-387 demonstrated strong antiviral activity against a range of different virus families, exceeding or matching the activity of known approved drug agents.
Superior to Other Treatments
NV-387 was substantially superior to remdesivir in coronavirus infections, using a model for SARS-CoV-2 (COVID) virus, as reported earlier. We believe that NV-387 continues to be one of the most active antiviral drugs against multiple coronaviruses, and that it is a viable clinical candidate for drug development to treat COVID, Long COVID, as well as potentially MERS, SARS, and seasonal coronavirus infections.
In treating Influenza, NV-387 was substantially superior to the three approved drugs, namely Tamiflu®, Rapivab® , and Xofluza® against an Influenza H3N2 lethal lung viral infection study, as previously reported. We believe that NV-387 is expected to possess strong antiviral activity against H5N1 "Bird Flu" as well, given that H5N1 viruses are known to bind to heparan sulfate proteoglycans, and based on the observed broad-spectrum activity of NV-387.
NNVC has also found that NV-387 is capable of completely curing a lethal RSV lung virus infection in animals, leading to indefinite survival of the animals, as reported recently. There is no cure for RSV, and no approved drug for treatment of RSV infection other than the toxic last-resort drug ribavirin.
Moreover, even novel viruses, whether from natural sources or bio-engineered, are expected to be susceptible to NV-387 if they employ SPG for gaining access to human cells to infect and cause disease. Thus, NV-387 could be highly valuable for preparedness against novel viral epidemics and pandemics.
NV-387 could thus be a single drug to treat all of the "tripledemic" viruses (COVID, RSV, FLU ), and more, when so approved!
Finally, NV-387 was at least as effective as the approved drug tecovitrimat (TPOXX®, SIGA), in a lethal intra-digital infection by ectromelia virus in mice. Importantly, a combined drug made from NV-387 and tecovirimat was more effective than either drug alone, indicating NV-387 "plays well" with tecovirimat and acts by a different mechanism.
- Smallpox poses a significant biodefense threat. Ectromelia virus is a native virus of mice in the poxvirus family and is one of the key animal model viruses for developing smallpox therapeutics. Tecovirimat is an approved drug for treating smallpox infection based on the FDA "Animal Rule", and is stockpiled by the US "Strategic National Stockpile". It was mobilized during the recent monkeypox epidemic.
- It is important to develop additional smallpox therapeutics that work well with tecovirimat and by themselves, since viruses pose the threat of drug escape by mutation; further, in a bio-terrorism scenario, a human-engineered smallpox virus resistant to existing drugs could be a potential threat.
Host-Mimetic Technology Solves the Greatest Challenge for Antiviral Medicines, i.e. Virus Escape!
No matter how much a virus changes in the field, it uses the same "landing sites" in the host body to gain access to cells, attach to and then fuse into the cells, causing an infection. A drug using the same landing sites and acting as a "decoy" would remain effective against the virus even as the virus changes to form variants, because the host side does not change.
NV-387 is designed to employ such advanced "host-mimetic" technology, built into the nanoviricide™ nanomedicine that is further designed to "look like a cell" to the virus.
In contrast, vaccines, antibodies and small chemical drugs all lose their effectiveness as the virus changes in the field. The virus is constantly changing due to various natural mechanisms such as mutations, recombinations, and/or re-assortments that are native to the virus life cycle.
Given the broad spectrum of antiviral activity of NV-387 against viruses in many different virus families, NNVC believes that its effectiveness against Influenza A/H3N2 is indicative of potential antiviral activity against most if not all Influenza A viruses.
Ultra-Broad-Spectrum Antiviral Nanomedicines Enabled by the Nanoviricides Platform Would Open Up Very Large Market Opportunities For The Company
Even with a decline since 2022, COVID-19 continues to hospitalize and kill people in the USA – the CDC website states 69,200 hospitalizations and 2,652 deaths since January 1, 2024; the worldwide market size for COVID-19 therapeutics is expected to exceed $16.2 Billion in 2031*.
*Source: Transparency Market Research
The market size for RSV therapeutics was estimated to be $2 Billion in 2023 and is expected to rise to exceed $8.5 Billion by the year 2031**.
**Source: Growth+ Market Reports.
NANOVIRICIDES VARIANT-PROOF, SAFE TECHNOLOGY THAT DOESN’T NEED THE HOST IMMUNE SYSTEM WILL TRANSFORM THE WAY VIRUSES & THEIR VARIANTS ARE TREATED WORLDWIDE!
NNVC’s novel approach has already enabled variant-proof drugs, blocking the complete viral life cycle without requiring help from the host’s defense systems! If both the viral re-infection cycle, and viral replication cycle arms of the viral lifecycle are blocked, a cure for many viral diseases is possible!!
NV-387 Possesses Strong Anti-Influenza-A Virus Activity, and May Also Have Activity Against H5N1 Bird Flu Virus
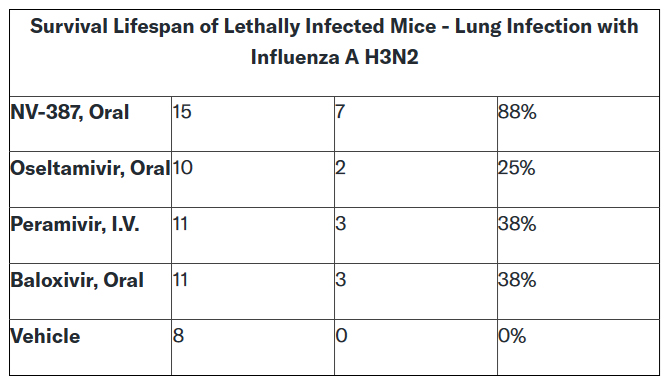
NNVC reported that in a lethal animal model of lung infection by Influenza A /H3N2 virus, NV-387 was found to have substantially superior antiviral effects compared to three approved anti-influenza drugs.
The company performed a lethal lung infection study of mice infected with Influenza A/H3N2 that were treated with NV-387 or one of the three approved drugs for direct comparison: Oseltamivir (Tamiflu®, Roche), Peramivir (Rapivab®, Biocryst), and Baloxivir (Xofluza®, Shionogi, Roche).
In this study, NV-387 Oral treatment led to a survival lifespan of 15 days, compared to 10 days with Oseltamivir Oral treatment, 11 days with Peramivir I.V. treatment, and 11 days with Baloxivir Oral treatment, while the vehicle-treated and untreated (infected) animals survived only 8 days!
Thus the anti-Influenza activity of NV-387 given orally was substantially superior to all three of the approved anti-influenza drugs, namely Tamiflu, Rapivab, and Xofluza.
Given the broad spectrum of antiviral activity of NV-387 against viruses in many different virus families, NNVC believes that its effectiveness against Influenza A/H3N2 is indicative of potential antiviral activity against most if not all Influenza A viruses.
Nanoviricides, Inc. is close to having a single drug NV-387 for the treatment of all of the "tripledemic" respiratory viruses - Coronaviruses, RSV, and Influenza A, which would be a revolutionary achievement!

Learn More about Nanoviricides, Inc. by gaining access to the latest corporate presentation
Download Research ReportNANOVIRICIDES are better because they destroy viruses and their variants without relying on the patient's immune system, thereby making them effective for populations that include geriatric and pediatric patients.
- Antibodies only bind by two points to the virus, and destruction of the complex requires effective immune function, which is not the case in sick patients..
- Vaccines only train the body into producing antibodies against the virus in the vaccine. Antibodies and vaccines are easily overcome by viruses by mutating in the field, hence the need for annual influenza vaccine updates.
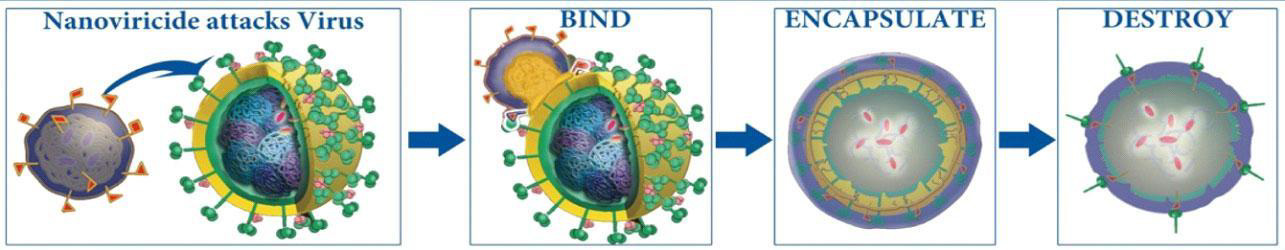
See Below for Nanoviricides' Unique, Novel, Post-Immunotherapeutic “Bind-Encapsulate-Destroy” Mechanism

THE BOTTOM LINE
With its ongoing expansion and development of their diverse and promising product pipeline, NanoViricides, Inc. (NYSE: NNVC) is positioned to be a dominant and innovative leader in the nanomedicine and antiviral therapy space.
NV-387 Could Revolutionize Antiviral Treatment Just as Antibiotics Did Against Bacteria!
The success of penicillin led to an explosion in development of antibacterials that use the same mechanism of action and provide additional benefits, stronger activity against certain bacteria and oral administration amongst them.
In fact, the world continues to use amoxicillin to treat most pediatric and adult bacterial infections to this day, some 65 years since its discovery in 1958! This durability is a direct result of the fact that most of the bacteria it attacks have not escaped the drug and have remained susceptible even after such a long time of common use.
NNVC has been able to develop NV-387 already for oral administration, as well as for injectable and inhalation formulations to enable many modes of use.
This comparison of features of nanoviricides technology platform and, in particular, NV-387, with the revolutionary antibiotics development against bacteria argues that NanoViricides (NYSE: NNVC) is now on a solid footing to revolutionize treatment of antiviral infections.
There is no denying that the world is changing… viruses are becoming more rampant. NNVC, an exciting NYSE company that is ahead of the problem, could see unstoppable growth ahead!
The company has sufficient funds to complete the on-going human clinical trials for its lead drug candidate NV-CoV-2 which is the drug product based on its "nanoviricide" active pharmaceutical ingredient ("API"), NV-387.
The successes of NV-387 as a broad-spectrum antiviral bode well for validating the multiple modalities in which NNVC’s Nanoviricides Platform Technology can be employed to revolutionize the treatment of viral infections as well as pandemic preparedness response.
This year the company has a focus on advancing NV-387 through phase II clinical trials of RSV, monkeypox/smallpox and other viruses including Flu/birdflu
NanoViricides, Inc.’s innovative platform technology, R&D agility, flexible cGMP compliant facility, highly effective management team, and extensive anti-viral experience should place this company at the top of your “invest” watchlist!

Learn More about Nanoviricides, Inc. by gaining access to the latest corporate presentation
Download PRESENTATIONTHIS IS A PAID ADVERTISEMENT
NO INVESTMENT ADVICE
SCD Media LLC (d/b/a “Smallcaps Daily”), hereinafter referred to as “Smallcaps Daily,” and their affiliates and control persons (the “Publisher”) are in the business of publishing favorable information and/or advertisements (the “Information”) about the securities of publicly traded companies (each an “Issuer” or collectively the “Issuers”) in exchange for compensation (the “Campaigns”). Persons receiving the Information are referred to as the “Recipients.” The person or entity paying the Publisher for the Campaign is referred to herein as the “Paying Party”. The Paying Party may be an Issuer, an affiliated or non-affiliate shareholder of an Issuer, or another person hired by the Issuer or an affiliate or non-affiliate shareholder of the Issuer. The nature and amount of compensation paid to the Publisher for the Campaign and creating and/or publishing the Information about each Issuer is set forth below under the heading captioned, “Compensation”.
This website provides information about the stock market and other investments. This website does not provide investment advice and should not be used as a replacement for investment advice from a qualified professional. This website is for informational purposes only. The Author of this website is not a registered investment advisor and does not offer investment advice. You, the reader, bear responsibility for your own investment decisions and should seek the advice of a qualified securities professional before making any investment.
Nothing on this website should be considered personalized financial advice. Any investments recommended herein should be made only after consulting with your personal investment advisor and only after performing your own research and due diligence, including reviewing the prospectus or financial statements of the issuer of any security.
Smallcaps Daily, its managers, its employees, affiliates, and assigns (collectively the "Publisher") do not make any guarantee or warranty about the advice provided on this website or what is otherwise advertised above.
Release of Liability: through use of this website, viewing or using you agree to hold Smallcaps Daily, its operators, owners, and employees harmless and to completely release them from any and all liability due to any and all loss (monetary or otherwise), damage (monetary or otherwise), or injury (monetary or otherwise) that you may incur. The information contained herein is based on sources that we believe to be reliable but is not guaranteed by us as being accurate and does not purport to be a complete statement or summary of the available data. Smallcaps Daily encourages readers and investors to supplement the information in these reports with independent research and other professional advice. All information on featured companies is provided by the company profiled or is available from public sources and Smallcaps Daily makes no representations, warranties, or guarantees as to the accuracy or completeness of the disclosure by the profiled company. None of the materials or advertisements herein constitute offers or solicitations to purchase or sell securities of the companies profiled herein and any decision to invest in any such company or other financial decisions should not be made based upon the information provided herein. Instead, Smallcaps Daily strongly urges you to conduct a complete and independent investigation of the respective companies and consideration of all pertinent risks. Smallcaps Daily’s full disclosure is to be read and fully understood before using Smallcaps Daily's website, or joining Smallcaps Daily's email or text list. From time to time, Smallcaps Daily will disseminate information about a company via website, email, sms, and other points of media. By viewing Smallcaps Daily's website and/or reading Smallcaps Daily's email or text newsletter you are agreeing to this ----> https://SmallcapsDaily.com/disclaimer/. All potential percentage gains discussed in any communications are based on calculations from the low to the high of the day. We are engaged in the business of marketing and advertising companies for monetary compensation.
If you have questions or concerns about a product you’ve seen in one of our emails, emails, text newsletters or SMS, we encourage you to reach out to that company directly.
Disclaimer – Always do your own research and consult with a licensed investment professional before investing. This communication is never to be used as the basis of making investment decisions and is for entertainment purposes only. At most, this communication should serve only as a starting point to do your own research and consult with a licensed professional regarding the companies profiled and discussed. Conduct your own research. This newsletter is a paid advertisement, not a recommendation nor an offer to buy or sell securities. This newsletter is owned, operated, and edited by the owner of Smallcaps Daily. Any wording found in this e-mail or disclaimer referencing to “I” or “we” or “our” refers to Smallcaps Daily. Our business model is to be financially compensated to market and promote small public companies. By reading our newsletter and our website you agree to the terms of our disclaimer, which are subject to change at any time. We are not registered or licensed in any jurisdiction whatsoever to provide investing advice or anything of an advisory or consultancy nature and are therefore unqualified to give investment recommendations. Companies with low prices per share are speculative and carry a high degree of risk, so only invest what you can afford to lose. By using our service, you agree not to hold our site, its editors, owners, or staff liable for any damages, financial or otherwise, that may occur due to any action you may take based on the information contained within our newsletters or on our website. We do not advise any reader to take any specific action. Losses can be larger than expected if the company experiences any problems with liquidity or wide spreads. Our website and newsletter are for entertainment purposes only. Never invest purely based on our alerts. Gains mentioned in our newsletter and on our website may be based on end-of-day or intraday data. This publication and its owners and affiliates may hold positions in the securities mentioned in our alerts, which we may sell at any time without notice to our subscribers, which may have a negative impact on share prices. If we own any shares, we will list the information relevant to the stock and the number of shares here.
COMPENSATION
In compliance with section 17(b) of the Securities Act we are disclosing that we have been compensated a fee pursuant to an agreement between Smallcaps Daily and IA Media LLC (d/b/a/ “IA Media”) hereinafter referred to as IA Media. Small Caps Daily was hired by IA Media for a period beginning January 2024 and ending January 2024 to publicly disseminate information about NanoViricides, Inc., via website, email, and SMS. We were paid up to twenty-five thousand usd via ACH. Subsequently, Small Caps Daily was hired by IA Media for a period beginning February 2024 and ending February 2024 to publicly disseminate information about NanoViricides, Inc., via website, email, and SMS. We were paid up to twenty-five thousand usd via ACH. Small Caps Daily was hired by IA Media for a period beginning July 1, 2025 and ending July 2, 2025 to publicly disseminate information about NanoViricides, Inc., via website, email, and SMS. We were paid up to twenty-five thousand usd via ACH.Readers are advised to review SEC periodic reports: forms 10Q 10K, form 8K, insider reports, forms 3, 4, 5 schedule 13d. Smallcaps Daily is compliant with the CAN-SPAM Act of 2003. Smallcaps Daily does not offer investment advice or analysis, and Smallcaps Daily further urges you to consult your own independent tax, business, financial, and investment advisors. investing in micro-cap, small-cap, and growth securities is highly speculative and carries an extremely high degree of risk. It is possible that an investor's investment may be lost or impaired due to the speculative nature of the companies profiled. The private securities litigation reform act of 1995 provides investors a safe harbor in regard to forward-looking statements. Any statements that express or involve discussions with respect to predictions, expectations, beliefs, plans, projections, objectives, goals, assumptions or future events, or performance are not statements of historical fact but may be forward-looking statements. Forward-looking statements are based on expectations, estimates, and projections at the time the statements are made that involve a number of risks and uncertainties which could cause actual results or events to differ materially from those presently anticipated. Forward-looking statements in this action may be identified through the use of words such as projects, foresee, expects, will, anticipates, estimates, believes, understands, or that by statements indicating certain actions & quotes; may, could, or might occur. Understand there is no guarantee past performance will be indicative of future results in preparing this publication. Smallcaps Daily has relied upon information supplied by its clients, as well as its clients’ publicly available information and press releases which it believes to be reliable; however, such reliability can not be guaranteed. Investors should not rely on the information contained on this website. Rather, investors should use the information contained in this website as a starting point for doing additional independent research on the featured companies. The advertisements in this website are believed to be reliable, however, Smallcaps Daily and its owners, affiliates, subsidiaries, officers, directors, representatives, and agents disclaim any liability as to the completeness or accuracy of the information contained in any advertisement and for any omissions of material facts from such advertisement. Smallcaps Daily is not responsible for any claims made by the companies advertised herein, nor is Smallcaps Daily responsible for any other promotional firm, its program, or its structure. Smallcaps Daily is not affiliated with any exchange, electronic quotation system, the Securities Exchange Commission, or FINRA.
Copyright © 2024 Smallcaps Daily. All rights reserved.
