Skye Bioscience (SKYE): A clinical stage biotech company working to unlock the pharmaceutical potential of the endocannabinoid system to treat several impactful diseases, including obesity

Renewed Pharma and institutional investor interest in CB1 receptors for weight loss could be a sign that underfollowed Skye Bioscience, Inc. (OTCQB: SKYE) is uniquely positioned in the rapidly growing obesity drug market
Skye is a biotech company developing proprietary molecules to treat diseases involving neurodegenerative, inflammatory, fibrotic, and metabolic conditions
The Company’s focus is on advancing first-in-class drugs with potential to provide new therapeutic outcomes for diseases with significant prevalence and unmet needs

Download Research Report
SKYE is unlocking the pharmaceutical potential of the endocannabinoid system (ECS) through modulation of the CB1 receptor
There is growing momentum in the endocannabinoid drug development frontier. Research suggests that the ECS may be a potential therapeutic target for numerous physiological conditions, including metabolic function and appetite regulation.
An opportunity revisited…
A promising class of obesity drugs - the cannabinoid receptor-1 inhibitor.
- Glaucoma
- Pain
- Cognitive function
- Neuroprotection
- Appetite regulation
- Weight loss
- Insulin sensitivity
- Kidney function
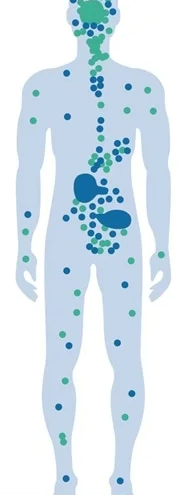
What do CB1 receptors do?
CB1 receptors make up one piece of a biological system called the endocannabinoid system (ECS). The ECS controls many basic biological processes, and exerts its activity in the nervous system and in the peripheral tissues, with direct involvement in food intake, energy balance, neuroprotection, pain control, sleep, mood, behavior and much more. As a consequence, ECS impairment has been reported in several diseases and its modulation represents therapeutic opportunities by targeting its receptors, such as CB1, for the treatment of multiple diseases.
How can CB1 receptors be influenced for therapeutic purposes?
While CB1 receptors are generally expressed in the brain and other parts of the central nervous system, there is convincing evidence of disorders in which the ECS, specifically the CB1 receptor, are upregulated in peripheral tissues in a manner that exacerbates certain disorders, such as obesity and kidney disease. As a result, blocking or inhibiting the CB1 receptor has been shown to have beneficial therapeutic effects. The most well known example of this was with a drug called rimonabant.

Years ago, CB1 receptors were looked at as potential therapeutic target for weight loss drugs. CB1-targeting drugs were poised to make a positive impact in this disease, until significant side effects derailed the commercialization of these drugs.
While Sanofi won clearance in Europe for its drug, rimonabant or Acomplia, it failed to gain support from the FDA’s outside advisors and was pulled from the European Union in 2009 over safety concerns. Because it acted on the brain, some of the side effects it caused were anxiety, depression, and suicidal thoughts.
Rimonabant had however demonstrated up to 10% weight loss over 1 year and improvements in metabolic outcomes.
Giants like Merck, Pfizer, Solvay, and other drug developers wound up sidelining similar assets over the side effect concern. This pullback by Big Pharma caused a setback in development of CB1 inhibitors.
More recently, a few innovative biotech companies developed new generations of CB1-targeting drugs, designing them with the goal of averting the detrimental effects in the brain. SKYE’s innovative technology is a differentiated approach that may overcome the key weakness of the 1st generation CB1 inhibitors to provide a safe and effective approach towards CB1 inhibition that could position CB1s to realize the therapeutic potential that their results suggested years ago.

With its leading effort to realize the pharmaceutical potential of endocannabinoid receptors, CB1 biotech company Skye Bioscience, Inc. (OTCQB: SKYE) is entering an important stage of its development.
With the potential of CB1 becoming clearer, distinct opportunities for SKYE are also emerging...
Company Overview:
Backed by leading biotechnology venture investors, SKYE’s strategy leverages biologic targets with substantial human proof of mechanism for the development of first-in-class therapeutics with significant clinical and commercial differentiation.
The company is focused on leveraging an important physiological system, the endocannabinoid system, that has only been recognized since the 1970s and is relatively underappreciated as a target for drug development.
SKYE aims to develop novel drugs with potential to treat glaucoma and other significant disease areas involving fibrotic, inflammatory and metabolic processes by targeting the CB1 receptor.

Why did specialized life science venture investors forge a deal to cannabinoid to endocannabinoid-targeting
Spurred by rising interest in molecules targeting endocannabinoid receptors to treat a range of diseases, specialized life science VCs joined forces with SKYE to develop therapies that target CB1. They facilitated a $20 million purchase that enabled Skye Bioscience Inc. (OTCQB:SKYE) to acquire Bird Rock Bio Inc’s nimacimab asset. They then invested $12 million in a PIPE deal to fund Skye’s operations and advance two molecules toward Phase II studies.
The deal set up SKYE to start its first Phase II study of its CB1 agonist/activator, SBI-100 OE, to treat glaucoma. It also allowed SKYE to start preparing its newly acquired Phase 2-ready drug nimacimab, a negative allosteric modulator of CB1, for a planned Phase 2 study for metabolic conditions with an emphasis on obesity and chronic kidney disease (CKD).
Studies have demonstrated a strong correlation between obesity and the development and progression of CKD. Obesity is intimately linked to hypertension and diabetes—the two biggest contributors to the development of kidney disease.
Nimacimab:
The company’s nimacimab is a peripherally acting CB1 inhibitor, a new approach to potentially address diseases such as obesity, pulmonary fibrotic diseases of the lungs, increased fat in the liver, and kidney function.
Early generations of CB1 inhibitors for weight loss acting on the central nervous system (CNS) showed efficacy but experienced setbacks, sometimes causing anxiety, depression, and suicidality.
As a monoclonal antibody, nimacimab does not easily cross what is called the “blood-brain barrier” and acts outside of the CNS. Animal toxicity studies showed no drug accumulation in the brain - a differentiating positive result.
- A Phase 1b study showed excellent safety with no adverse events of concern and encouraging trends in exploratory biomarkers after three-week dosing.
- Skye is planning to start a Phase 2 study focused on an obesity and chronic kidney disease in H1 2024.
Establishing proof-of-concept could pave the way to potentially address multiple diseases with significant prevalence and unmet need.

SKYE’s nimacimab, a next-generation CB1 inhibitor, could be a promising solution to help address obesity: a rapidly growing condition with notably detrimental health effects.
The impact of drugs like Ozempic and Wegovy has made numerous headlines in 2023. Celebrities, athletes, and regular people have been turning to these drugs to lose weight.
Semaglutide falls under a drug class called GLP-1 receptor agonists, which attach to GLP-1 receptors in the body and produce more insulin, helping to lower blood sugar levels and decrease appetite.
U.S. doctors have been writing out millions of prescriptions for obesity drugs including semaglutide (sold under the brand names Ozempic, Wegovy) in the last year. This growth in prescriptions has translated into rapid sales growth for the two pharmaceutical company leaders in this space, Novo Nordisk and Eli Lilly.
But there’s a problem….
Studies have found that semaglutide leads to a significant decrease in fat-free mass (aka muscle mass) compared with a placebo. Furthermore, because this drug is focused on reducing appetite, if patients go off the drug their food consumption increases and they readily regain weight.
SKYE’s nimacimab uses a different mechanism with the goal of achieving important, long-lasting impacts on weight and related conditions such as kidney diseases. Biotech observers believe there are numerous opportunities for differentiated and improved therapeutic outcomes in the burgeoning weight loss and cardiometabolic space. If nimacimab can build on the positive prior evidence of CB1 inhibition in its upcoming clinical trial and beyond, there is a clear opportunity for it to play a distinct role in this emerging future.

About SBI-100 Ophthalmic Emulsion:
Skye's SBI-100 OE possesses a novel molecular structure and nanoemulsion formulation that were designed to enable effective topical delivery and better penetration of a CB1 agonist into ocular tissue.
In preclinical studies involving three different species, the drug resulted in enhanced therapeutic efficacy and duration of response in lowering intraocular pressure, comparing favorably to the standard of care for treating glaucoma.
The company recently announced successful results from its Phase 1 clinical trial assessing the safety and pharmacokinetics of SBI-100 Ophthalmic Emulsion ("OE").
This first-in-class cannabinoid type 1 receptor ("CB1") agonist is a prodrug uniquely formulated as an eyedrop and is being developed as a potential new mechanism to address unmet needs in the treatment of glaucoma.
"Our Skye team is proud to have completed our first human trial of an ophthalmic drug targeting the CB1 receptor and accomplishing a major step on our journey to provide ophthalmology healthcare professionals, and their glaucoma patients, access to a new class of medicine. There remain notable unmet needs in the treatment regimen for glaucoma. A notable portion of glaucoma patients fail their initial prescribed medications. In addition, hyperaemia, or red eyes, is a common side effect of glaucoma drugs and is a major reason for patients discontinuing these treatments. We believe that SBI-100 OE has a distinct potential role to play as a new mechanism for this disease."
Punit Dhillon, CEO and Chair of Skye
Top Reasons to Have SKYE on Your Radar:
- Big pharma is seeing the appeal of CB1 receptors: Recent acquisitions highlight interest in the ECS: Jazz Pharmaceuticals/GW Pharmaceuticals/epilepsy (2021); Horizon/Zynerba/Fragile X drug (2023); in 2023, the largest seller of drugs used for weight loss, Novo Nordisk, acquired Inversago Pharma and its peripheral CB1-inhibiting drug (with Phase 1 data).
- Experienced leadership: Contributed to the commercialization of 47+ drugs/diagnostics, led high-value strategic transactions, and co-founded multiple companies.
- Promising outlook: The company has strong execution, a strong track record, and a clear clinical path forward.
- Growing market for obesity drugs: Wegovy and Ozempic sales have ballooned in recent years and the company has a product that could potentially play a vital role as a single or combination drug in this market - one that may not cause muscle loss or CNS liabilities!
- High unmet need among glaucoma patients. Eyecare providers are showing a high level of familiarity and enthusiasm for a pharmaceutical-quality CB1 agonist as a new class of drug for glaucoma.
In Summary….
Focusing on the endocannabinoid system (ECS) may be the answer to the development of improved drugs for key diseases. This may be game-changing for SKYE as it advances through the clinic with a potential new drug in the obesity and glaucoma markets.
The success of new drugs for obesity is creating even greater enthusiasm for and investment in drugs that may further advance their benefit for weight loss and other related cardiometabolic conditions, and also potentially create opportunities for combinations with the current leading drugs, Wegovy and Ozempic. This in turn is creating an important opportunity for Skye to advance its unique drug, nimacimab.
Backed by leading biotech venture investors, Skye Bioscience, Inc. (OTC: SKYE) is spearheading its own distinct approach to realize the potential for CB1 receptors to positively impact the medical landscape.
Learn More about Skye Bioscience, Inc. by gaining access to their comprehensive investor presentation
Download Report
Contact us
SmallCapsDaily, LLC
1334 Northampton St, Easton, PA 18042
info@smallcapsdaily.com
THIS IS A PAID ADVERTISEMENT
NO INVESTMENT ADVICE
Forward looking statements disclaimer
Skye Safe Harbor Statement
The content of this landing page contains forward-looking statements, including statements regarding our product development, business strategy, timing of clinical trials and commercialization of cannabinoid-derived therapeutics. Such statements and other statements in this press release that are not descriptions of historical facts are forward-looking statements that are based on management's current expectations and assumptions and are subject to risks and uncertainties. If such risks or uncertainties materialize or such assumptions prove incorrect, our business, operating results, financial condition, and stock price could be materially negatively affected. In some cases, forward-looking statements can be identified by terminology including "anticipated," "plans," "goal," "focus," "aims," "intends," "believes," "can," "could," "challenge," "predictable," "will," "would," "may" or the negative of these terms or other comparable terminology. We operate in a rapidly changing environment and new risks emerge from time to time. As a result, it is not possible for our management to predict all risks, nor can we assess the impact of all factors on our business or the extent to which any factor, or combination of factors, may cause actual results to differ materially from those contained in any forward-looking statements the Company may make. Risks and uncertainties that may cause actual results to differ materially include, among others, our capital resources, uncertainty regarding the results of future testing and development efforts and other risks that are described in the Risk Factors section of Skye's most recent annual or quarterly report filed with the Securities and Exchange Commission. Except as expressly required by law, Skye disclaims any intent or obligation to update these forward-looking statements. Skye Bioscience has entered into an agreement to pay Tradigital Marketing Group an amount of $32,500 for digital marketing and advertising services on a consulting basis and has agreed to also pay to Interactive Offers,LLC a digital advertising traffic acquisition cost of up to one hundred $112,500.
SCD Media LLC (d/b/a “Smallcaps Daily”), hereinafter referred to as “Smallcaps Daily,” and their affiliates and control persons (the “Publisher”) are in the business of publishing favorable information and/or advertisements (the “Information”) about the securities of publicly traded companies (each an “Issuer” or collectively the “Issuers”) in exchange for compensation (the “Campaigns”). Persons receiving the Information are referred to as the “Recipients.” The person or entity paying the Publisher for the Campaign is referred to herein as the “Paying Party”. The Paying Party may be an Issuer, an affiliated or non-affiliate shareholder of an Issuer, or another person hired by the Issuer or an affiliate or non-affiliate shareholder of the Issuer. The nature and amount of compensation paid to the Publisher for the Campaign and creating and/or publishing the Information about each Issuer is set forth below under the heading captioned, “Compensation”.
This website provides information about the stock market and other investments. This website does not provide investment advice and should not be used as a replacement for investment advice from a qualified professional. This website is for informational purposes only. The Author of this website is not a registered investment advisor and does not offer investment advice. You, the reader, bear responsibility for your own investment decisions and should seek the advice of a qualified securities professional before making any investment.
Nothing on this website should be considered personalized financial advice. Any investments recommended herein should be made only after consulting with your personal investment advisor and only after performing your own research and due diligence, including reviewing the prospectus or financial statements of the issuer of any security.
Smallcaps Daily, its managers, its employees, affiliates, and assigns (collectively the "Publisher") do not make any guarantee or warranty about the advice provided on this website or what is otherwise advertised above.
Release of Liability: through use of this website, viewing or using you agree to hold Smallcaps Daily, its operators, owners, and employees harmless and to completely release them from any and all liability due to any and all loss (monetary or otherwise), damage (monetary or otherwise), or injury (monetary or otherwise) that you may incur. The information contained herein is based on sources that we believe to be reliable but is not guaranteed by us as being accurate and does not purport to be a complete statement or summary of the available data. Smallcaps Daily encourages readers and investors to supplement the information in these reports with independent research and other professional advice. All information on featured companies is provided by the company profiled or is available from public sources and Smallcaps Daily makes no representations, warranties, or guarantees as to the accuracy or completeness of the disclosure by the profiled company. None of the materials or advertisements herein constitute offers or solicitations to purchase or sell securities of the companies profiled herein and any decision to invest in any such company or other financial decisions should not be made based upon the information provided herein. Instead, Smallcaps Daily strongly urges you to conduct a complete and independent investigation of the respective companies and consideration of all pertinent risks. Smallcaps Daily’s full disclosure is to be read and fully understood before using Smallcaps Daily's website, or joining Smallcaps Daily's email or text list. From time to time, Smallcaps Daily will disseminate information about a company via website, email, sms, and other points of media. By viewing Smallcaps Daily's website and/or reading Smallcaps Daily's email or text newsletter you are agreeing to this ----> https://Smallcaps Daily.com/disclaimer/. All potential percentage gains discussed in any communications are based on calculations from the low to the high of the day. We are engaged in the business of marketing and advertising companies for monetary compensation.
If you have questions or concerns about a product you’ve seen in one of our emails, emails, text newsletters or SMS, we encourage you to reach out to that company directly.
Disclaimer – Always do your own research and consult with a licensed investment professional before investing. This communication is never to be used as the basis of making investment decisions and is for entertainment purposes only. At most, this communication should serve only as a starting point to do your own research and consult with a licensed professional regarding the companies profiled and discussed. Conduct your own research. This newsletter is a paid advertisement, not a recommendation nor an offer to buy or sell securities. This newsletter is owned, operated, and edited by the owner of Smallcaps Daily. Any wording found in this e-mail or disclaimer referencing to “I” or “we” or “our” refers to Smallcaps Daily. Our business model is to be financially compensated to market and promote small public companies. By reading our newsletter and our website you agree to the terms of our disclaimer, which are subject to change at any time. We are not registered or licensed in any jurisdiction whatsoever to provide investing advice or anything of an advisory or consultancy nature and are therefore unqualified to give investment recommendations. Companies with low prices per share are speculative and carry a high degree of risk, so only invest what you can afford to lose. By using our service, you agree not to hold our site, its editors, owners, or staff liable for any damages, financial or otherwise, that may occur due to any action you may take based on the information contained within our newsletters or on our website. We do not advise any reader to take any specific action. Losses can be larger than expected if the company experiences any problems with liquidity or wide spreads. Our website and newsletter are for entertainment purposes only. Never invest purely based on our alerts. Gains mentioned in our newsletter and on our website may be based on end-of-day or intraday data. This publication and its owners and affiliates may hold positions in the securities mentioned in our alerts, which we may sell at any time without notice to our subscribers, which may have a negative impact on share prices. If we own any shares, we will list the information relevant to the stock and the number of shares here.
COMPENSATION
In compliance with section 17(b) of the Securities Act Small Caps Daily is disclosing that we have been compensated a fee pursuant to an agreement between Smallcaps Daily and Interactive Offers LLC. Small Caps Daily was paid up to twenty-five hundred usd via ACH for web hosting this landing page. Readers are advised to review SEC periodic reports: forms 10Q 10K, form 8K, insider reports, forms 3, 4, 5 schedule 13d. Smallcaps Daily is compliant with the CAN-SPAM Act of 2003. Smallcaps Daily does not offer investment advice or analysis, and Smallcaps Daily further urges you to consult your own independent tax, business, financial, and investment advisors. Investing in micro-cap, small-cap, and growth securities is highly speculative and carries an extremely high degree of risk. It is possible that an investor's investment may be lost or impaired due to the speculative nature of the companies profiled. The private securities litigation reform act of 1995 provides investors a safe harbor in regard to forward-looking statements. Any statements that express or involve discussions with respect to predictions, expectations, beliefs, plans, projections, objectives, goals, assumptions or future events, or performance are not statements of historical fact but may be forward-looking statements. Forward-looking statements are based on expectations, estimates, and projections at the time the statements are made that involve a number of risks and uncertainties which could cause actual results or events to differ materially from those presently anticipated. Forward-looking statements in this action may be identified through the use of words such as projects, foresee, expects, will, anticipates, estimates, believes, understands, or that by statements indicating certain actions & quotes; may, could, or might occur. Understand there is no guarantee past performance will be indicative of future results in preparing this publication. Smallcaps Daily has relied upon information supplied by its clients, as well as its clients’ publicly available information and press releases which it believes to be reliable; however, such reliability can not be guaranteed. Investors should not rely on the information contained on this website. Rather, investors should use the information contained in this website as a starting point for doing additional independent research on the featured companies. The advertisements in this website are believed to be reliable, however, Smallcaps Daily and its owners, affiliates, subsidiaries, officers, directors, representatives, and agents disclaim any liability as to the completeness or accuracy of the information contained in any advertisement and for any omissions of material facts from such advertisement. Smallcaps Daily is not responsible for any claims made by the companies advertised herein, nor is Smallcaps Daily responsible for any other promotional firm, its program, or its structure. Smallcaps Daily is not affiliated with any exchange, electronic quotation system, the Securities Exchange Commission, or FINRA.
Copyright © 2022 Smallcaps Daily. All rights reserved.
