This Undiscovered Biotech Has Blockbuster Potential with the Only Fully Human anti-CD3 Monoclonal Antibody in Clinical Development and Sanofi Just Invested Heavily in the Peer Group Through the Acquisition of Provention Bio for $2.9 BILLION DOLLARS…

Introducing Tiziana Life Sciences (NASDAQ: TLSA): a Clinical Stage Biotech Company with a Unique Approach that Specializes in the Developing of Transformative Therapies for Neurodegenerative and Lung Diseases.
Historically, monoclonal antibodies, such as blockbuster drug, Humira®, have performed as the best selling drugs in the world. Targeting across multi-indications such as autoimmune disorders and degenerative diseases, these drugs have multi-billion dollar blockbuster potential.
To this day, there is no drug on the market that is a fully human, anti-CD3 monoclonal antibody…and Tiziana Life Sciences (NASDAQ: TLSA) is developing the potential to treat patients in need that suffer from secondary progressive multiple sclerosis.

Download Research Report
Tiziana Life Sciences (NASDAQ: TLSA)’s drug, foralumab, is the world’s only fully human anti-CD3 monoclonal antibody in clinical development
Tiziana’s mission is to bring breakthrough therapies to patients with the aim of treating Secondary Progressive Multiple Sclerosis, ALS, Alzheimer's, and other CNS indications, Crohn's Disease, lung diseases and optimizing health outcomes.
Tiziana Life Sciences is developing a transformational intranasal formulation, switching from traditional routes to alternative routes to enable breakthrough immunomodulation approaches to enhance the functionality of Treg-based therapies.

Tiziana Life Sciences (NASDAQ: TLSA) has the potential to change the way immunotherapies are currently conducted with their revolutionary approach to attacking neurodegenerative and lung diseases
Tiziana Life Sciences (NASDAQ: TLSA) is Gaining Global Recognition for its First In Class asset, foralumab, a unique Intranasal monoclonal antibody:
- Forbes Article: March 16, 2023 - “New T Cell Antibody Treatment Improves Outcomes For Covid Patients”
- PNAS Highlight: https://www.pnas.org/
-
- Jama Network Article: Published March 22, 2023: https://jamanetwork.com/journals/jama/fullarticle/2802889
Market Potential:
Neurodegenerative diseases such as Multiple Sclerosis and Alzheimer’s have risen globally due to the aging population. The NIH projects that in the United States, as many as 6.2 million people may have Alzheimer’s disease.
Neurodegenerative diseases occur when nerve cells in the brain or peripheral nervous system lose function over time and ultimately die. Treatments may help relieve some of the physical or mental symptoms associated with neurodegenerative diseases, however, slowing their progression is not currently possible, and no cures exist.
This rising problem is driving market growth with the neurodegenerative diseases market projected to reach USD $53 Billion by 2030 and grow steadily at a CAGR of 3.2%.
Currently, there are only three measures the FDA considers validated for Multiple Sclerosis and Tiziana Life Sciences is studying all of them
With an impressive team and board of scientists, Tiziana Life Sciences aims to disrupt biopharma with their potentially breakthrough therapies to patients in need
Investment Highlights:
A revolutionary platform: TLSA is first in class, switching from uncomfortable antibody injections to revolutionary nasal and oral methods of delivery. The benefits of switching to this unique methodology are increased patient compliance, minimized side effects, and anticipated lower cost of goods and administration.
Multiple Indications: TLSA’s focus is across multiple indications such as neurodegenerative diseases like Multiple Sclerosis as well as lung diseases. Similar monoclonal antibody drugs, such as Humira®, have performed extraordinarily well with approximately $200 billion dollars in revenue.
Monoclonal Antibodies: Recently, Provention Bio, a competitor for TLSA, was acquired by Sanofi SA (NASDAQ: SNY) for $2.9 billion. Tiziana stands out from the competition as their drug, foralumab, is administered nasally and is fully human.
Foralumab: Tiziana’s clinical drug, foralumab, a fully human anti-CD3 monoclonal antibody for the treatment of neurodegenerative diseases, works with novel technology to be delivered nasally. This allows the drug to bypass and potentially prevent the toxicities that may be present with non-fully human antibodies making it a revolutionary and very exciting clinical development.
Market Potential: Neurodegenerative diseases are rising which contributes to a growing market market projected to reach USD 53 Billion by 2030 and grow steadily at a CAGR of 3.2%. Beyond that, competitors are catching the attention of big pharma.
Unmet Need: TLSA is dedicated to developing drugs to service diseases that currently have no cure. The company’s clinical trial is across multiple neurodegenerative disease, autoimmune, and lung disease indications.
Greetings Investors,
Even in our day and age, there is currently still no cure for neurodegenerative diseases such as Alzheimer's and ALS. The rise of diagnoses can be attributed to the aging population as life expectancy increases.
Neurodegenerative diseases collectively affected 4.7 to 6.0 million individuals in the U.S. between 2016-2017. These diseases were responsible for 272,644 deaths and 3,011,484 disability adjusted life years in 2016.
Not only is this a growing health concern, but the annual cost to the US from Alzheimer’s disease and related dementias and other neurodegenerative diseases was $655 billion in 2020 USD.
Autoimmune diseases are conditions in which your immune system mistakenly attacks your body. A healthy immune system defends the body against disease and infection. But if the immune system malfunctions, it mistakenly attacks healthy cells, tissues, and organs. These attacks can affect any part of the body, weakening bodily function and even become life-threatening. These diseases affect more than 24 million people in the United States.
Now more than ever we are in need of companies like Tiziana Life Sciences, whose focus is on developing drugs with a unique approach for these hard to treat but rising diseases.
TLSA is revolutionary in the biotech space as the company works to deliver on its clinical strategy through an exciting pipeline with drug development programs across several degenerative and autoimmune disease indications that have a large unmet need
Company Overview
Tiziana Life Sciences (NASDAQ: TLSA) is a clinical stage biotechnology company that specializes in developing transformative therapies for neurodegenerative and lung diseases. The Company’s clinical pipeline includes drug assets for Secondary Progressive Multiple Sclerosis, ALS, Alzheimer's, Crohn's Disease and KRAS+ NSCLC.
Tiziana is led by a team of highly qualified executives with extensive drug development and commercialization experience.The Company is developing transformational formulation technologies, enabling it to switch from traditional routes to alternative routes of immunotherapy to facilitate local sites of action.

Strategy
- Continue the clinical development of intranasal administration of foralumab for neurodegenerative disease indications:
- Secondary Progressive, Multiple Sclerosis
- Alzheimer's
- Amyotrophic Lateral Sclerosis (ALS)
- Submit an Investigational New Drug Application (IND) to conduct a Phase 2 study intranasal foralumab in Alzheimer's disease patients.
- Seek orphan drugs fast track or breakthrough designation for our product candidates where warranted.

Foraluamb
Foralumab is the Only Fully Human anti-CD3 mAb in Clinical Trials
CD3-specific Monoclonal Antibodies in Clinical Development
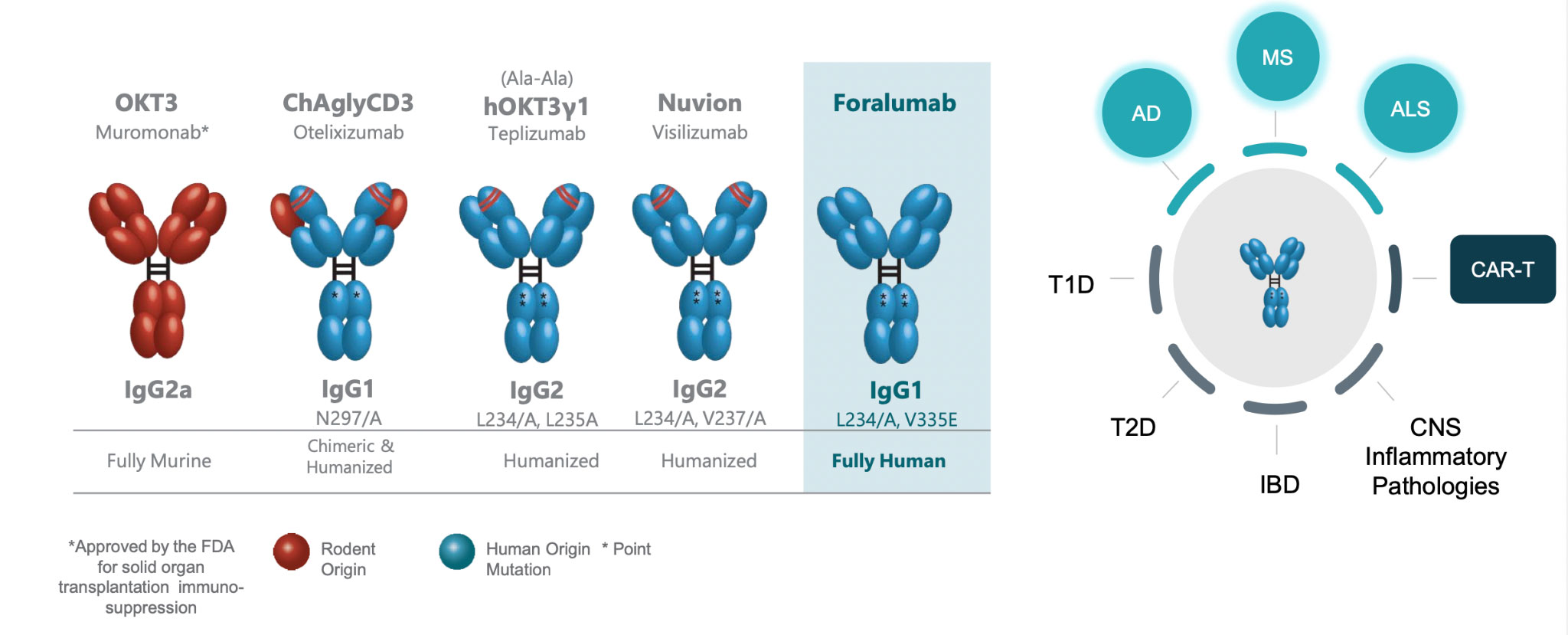
Foralumab is a fully human anti-CD3 monoclonal antibody (mAb) for the treatment of Crohn’s and neurodegenerative diseases. This is administered to mucosal surfaces in the nose (intranasal) to boost regulatory T-cell (T-reg) responses with the aim of controlling autoimmune disease progression in central nervous system (CNS) indications like multiple sclerosis (MS) and Alzheimer's disease (AD).
Tiziana has recently completed its Phase 1 clinical trial using intranasal foralumab and is poised to start Phase 2 for its non-active secondary progressive MS indication with nasal administration in Q3 2023.
Intranasally Administered Foralumab in Non-Active SPMS Patients
Two patients were dosed with intranasal foralumab M-W-F for two weeks with a subsequent 1-week washout period (regimen then repeated).
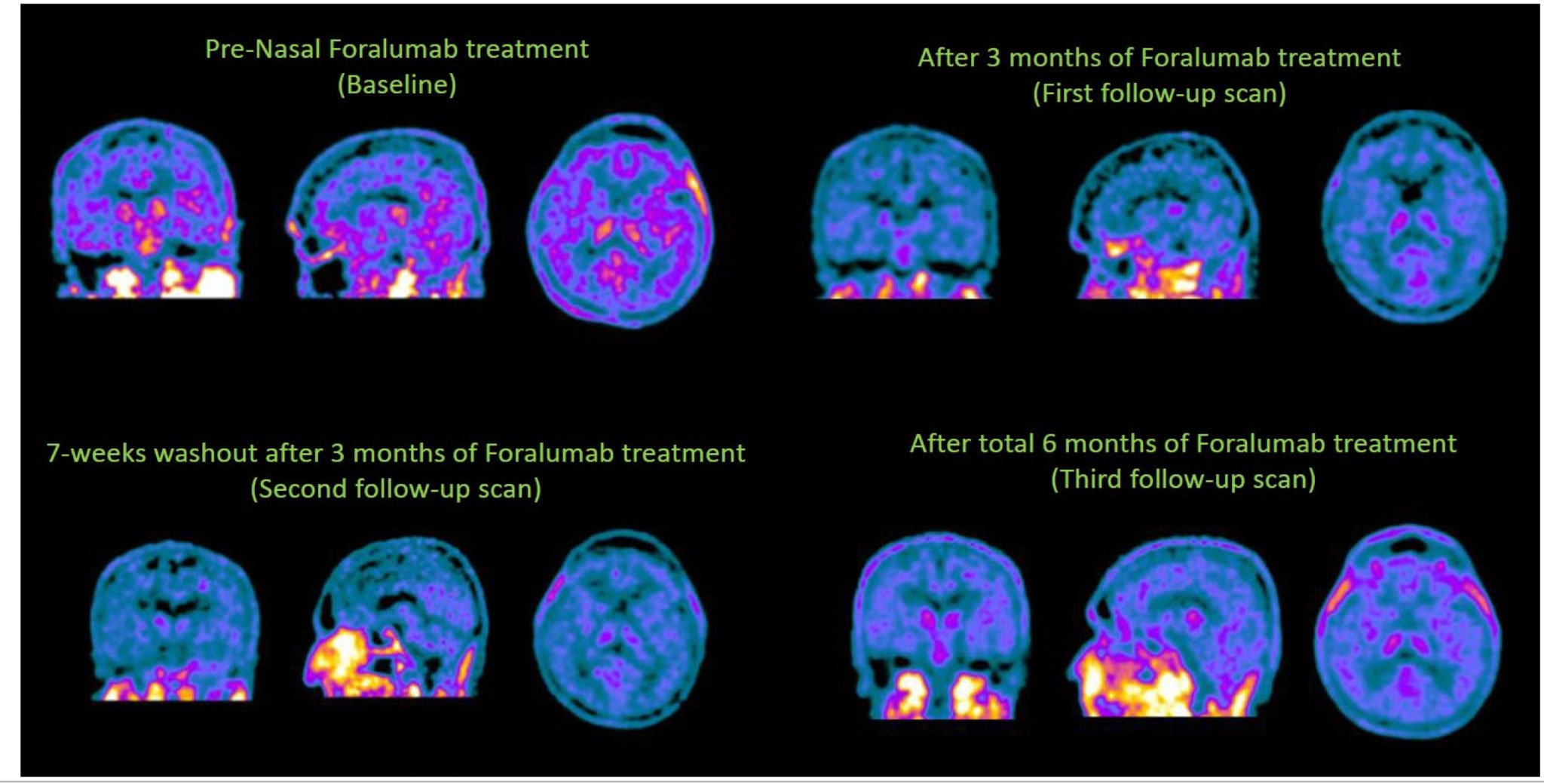
Positive Results: The regimen was well-tolerated with associated beneficial clinical and biomarker changes.
Foralumab has demonstrated ability to activate regulatory T cells that systemically circulate to elicit targeted immunomodulation providing therapeutic benefit to patients.
Tiziana has submitted a patent application on potential use of Foralumab, to improve success of chimeric antigen receptor T cells (CAR-T) therapy for cancer and other human diseases.

Gabriele Cerrone
Executive Chairman Mr Cerrone has a successful track record and extensive experience in the financing and restructuring of micro-cap biotechnology companies. He has founded nine biotechnology companies in oncology, infectious diseases and molecular diagnostics, and has taken six of these companies to the NASDAQ Market and one to the AIM Market in London. Mr Cerrone co-founded Trovagene, Inc. (NASDAQ: TROV), a molecular diagnostic company and served as its Co-Chairman; he was a co-founder and served as Chairman of both Synergy Pharmaceuticals, Inc. (NASDAQ: SGYP) and Callisto Pharmaceuticals, Inc. (OTCMKTS: CLSP), and was a Director of and led the restructuring of Siga Technologies, Inc. (NASDAQ: SIGA). Mr Cerrone also co-founded FermaVir Pharmaceuticals, Inc. and served as Chairman of the Board until its merger in September 2007 with Inhibitex, Inc. Mr Cerrone served as a director of Inhibitex, Inc. until its US$2.5bn sale to Bristol Myers Squibb Co in 2012. Mr Cerrone is the Executive Chairman and Co-Founder of Gensignia Life Sciences, Inc., a molecular diagnostics company focused on oncology using microRNA technology; Chairman and Founder of Tiziana Life Sciences plc (AIM: TILS) an oncology focused therapeutics company; Chairman and Co-Founder of Rasna Therapeutics Limited, a company focused on the development of therapeutics for leukaemias; Co-Founder of ContraVir Pharmaceuticals, Inc. (Nasdaq: CTRV); and founder of BioVitas Capital Ltd.
Matthew W Davis, MD, RPh
Chief Medical Officer and acting Chief Scientific Officer Dr. Davis has extensive experience in new drug application, or NDA, and biologic license application, or BLA, FDA approvals and device clearances. Notable approved brands that Dr. Davis has worked on include Lidoderm®, Sculptra®, Colcrys® and most recently QWO®. Dr. Davis previously served as Chief Scientific Officer and Chief Medical Officer at Endo Pharmaceuticals where he restructured the R&D department and collaborated to obtain BLA approval for QWO®. Additionally, Dr. Davis was Chief Medical Officer for Lupin Inc. and URL Pharma, Inc. where he spearheaded three NDA approvals and was the inventor on all 17 Orange Book listed patents for Colcrys®. He also was on the executive team that sold URL Pharma to Takeda Pharmaceutical Company for approximately $800M combined with over $1B in performance-based contingent earn out payments. Dr. Davis matriculated to the University of Pennsylvania as an undergraduate. He received his Pharmacy Degree from Temple University and his Medical Degree from the Medical College of Pennsylvania. Dr. Davis undertook his surgical training at Brown University and his Urology training at Washington Hospital Center
Keeren Shah
Chief Financial Officer Keeren Shah serves as our Chief Financial Officer. Ms. Shah currently also serves as the Chief Financial Officer of OKYO Pharma Ltd, Accustem Sciences Limited and Rasna Therapeutics Inc., having previously served as the Group Financial Controller for all businesses from June 2016 to July 2020. Prior to joining the Company, Ms. Shah spent 10 years at Visa, Inc. as a Senior Leader in its finance team where she was responsible for key financial controller activities, financial planning and analysis, and core processes as well as leading and participating in key transformation programmes and Visa Inc.’s initial public offering. Before joining Visa, Ms. Shah also held a variety of finance positions at other leading companies including Arthur Andersen and BBC Worldwide. She holds a Bachelor of arts with honors in Economics and is a member of the Chartered Institute of Management Accountants.In Conclusion
Tiziana Life Sciences (NASDAQ: TLSA) is a revolutionary, clinical stage biotechnology company that specializes in developing transformative therapies for neurodegenerative and lung diseases. The Company is gaining global attention in the media, and was most recently (March, 2023) highlighted in Forbes and Bloomberg.
Most excitingly, the Company’s furthest advanced clinical trial is centered on Foralumab, which is the world’s only fully human anti-CD3 monoclonal antibody for the treatment of Crohn’s and neurodegenerative diseases, and the first administered intranasally. Investors should take note of M&A in the space with a close peer, Provention Bio Inc. (Nasdaq: PRVB), which was recently acquired by Sanofi for $2.9B.
Other blockbuster monoclonal drugs such as Humira, have performed as the best selling drugs in the world with $200 billion in revenue. Big Pharma is paying attention to these drugs and TLSA is standing out from the competition with its unique application through nasal inhalation.
TLSA is dedicated to developing drugs to service diseases that currently have no cure but a major medical need. With three clinical trials across neurodegenerative, autoimmune, and lung disease indications, the company is attacking multiple indications.
Tiziana Life Sciences (NASDAQ: TLSA) is a first in class biotechnology company with the world’s only fully human anti-CD3 monoclonal antibody in clinical development making the company a very exciting player to have on your watch list
Start your research today.

THIS IS A PAID ADVERTISEMENT
NO INVESTMENT ADVICE
SCD Media LLC (d/b/a “Smallcaps Daily”), hereinafter referred to as “Smallcaps Daily,” and their affiliates and control persons (the “Publisher”) are in the business of publishing favorable information and/or advertisements (the “Information”) about the securities of publicly traded companies (each an “Issuer” or collectively the “Issuers”) in exchange for compensation (the “Campaigns”). Persons receiving the Information are referred to as the “Recipients.” The person or entity paying the Publisher for the Campaign is referred to herein as the “Paying Party”. The Paying Party may be an Issuer, an affiliated or non-affiliate shareholder of an Issuer, or another person hired by the Issuer or an affiliate or non-affiliate shareholder of the Issuer. The nature and amount of compensation paid to the Publisher for the Campaign and creating and/or publishing the Information about each Issuer is set forth below under the heading captioned, “Compensation”.
This website provides information about the stock market and other investments. This website does not provide investment advice and should not be used as a replacement for investment advice from a qualified professional. This website is for informational purposes only. The Author of this website is not a registered investment advisor and does not offer investment advice. You, the reader, bear responsibility for your own investment decisions and should seek the advice of a qualified securities professional before making any investment.
Nothing on this website should be considered personalized financial advice. Any investments recommended herein should be made only after consulting with your personal investment advisor and only after performing your own research and due diligence, including reviewing the prospectus or financial statements of the issuer of any security.
Smallcaps Daily, its managers, its employees, affiliates, and assigns (collectively the "Publisher") do not make any guarantee or warranty about the advice provided on this website or what is otherwise advertised above.
Release of Liability: through use of this website, viewing or using you agree to hold Smallcaps Daily, its operators, owners, and employees harmless and to completely release them from any and all liability due to any and all loss (monetary or otherwise), damage (monetary or otherwise), or injury (monetary or otherwise) that you may incur. The information contained herein is based on sources that we believe to be reliable but is not guaranteed by us as being accurate and does not purport to be a complete statement or summary of the available data. Smallcaps Daily encourages readers and investors to supplement the information in these reports with independent research and other professional advice. All information on featured companies is provided by the company profiled or is available from public sources and Smallcaps Daily makes no representations, warranties, or guarantees as to the accuracy or completeness of the disclosure by the profiled company. None of the materials or advertisements herein constitute offers or solicitations to purchase or sell securities of the companies profiled herein and any decision to invest in any such company or other financial decisions should not be made based upon the information provided herein. Instead, Smallcaps Daily strongly urges you to conduct a complete and independent investigation of the respective companies and consideration of all pertinent risks. Smallcaps Daily’s full disclosure is to be read and fully understood before using Smallcaps Daily's website, or joining Smallcaps Daily's email or text list. From time to time, Smallcaps Daily will disseminate information about a company via website, email, sms, and other points of media. By viewing Smallcaps Daily's website and/or reading Smallcaps Daily's email or text newsletter you are agreeing to this ----> https://Smallcaps Daily.com/disclaimer/. All potential percentage gains discussed in any communications are based on calculations from the low to the high of the day. We are engaged in the business of marketing and advertising companies for monetary compensation.
If you have questions or concerns about a product you’ve seen in one of our emails, emails, text newsletters or SMS, we encourage you to reach out to that company directly.
Disclaimer – Always do your own research and consult with a licensed investment professional before investing. This communication is never to be used as the basis of making investment decisions and is for entertainment purposes only. At most, this communication should serve only as a starting point to do your own research and consult with a licensed professional regarding the companies profiled and discussed. Conduct your own research. This newsletter is a paid advertisement, not a recommendation nor an offer to buy or sell securities. This newsletter is owned, operated, and edited by the owner of Smallcaps Daily. Any wording found in this e-mail or disclaimer referencing to “I” or “we” or “our” refers to Smallcaps Daily. Our business model is to be financially compensated to market and promote small public companies. By reading our newsletter and our website you agree to the terms of our disclaimer, which are subject to change at any time. We are not registered or licensed in any jurisdiction whatsoever to provide investing advice or anything of an advisory or consultancy nature and are therefore unqualified to give investment recommendations. Companies with low prices per share are speculative and carry a high degree of risk, so only invest what you can afford to lose. By using our service, you agree not to hold our site, its editors, owners, or staff liable for any damages, financial or otherwise, that may occur due to any action you may take based on the information contained within our newsletters or on our website. We do not advise any reader to take any specific action. Losses can be larger than expected if the company experiences any problems with liquidity or wide spreads. Our website and newsletter are for entertainment purposes only. Never invest purely based on our alerts. Gains mentioned in our newsletter and on our website may be based on end-of-day or intraday data. This publication and its owners and affiliates may hold positions in the securities mentioned in our alerts, which we may sell at any time without notice to our subscribers, which may have a negative impact on share prices. If we own any shares, we will list the information relevant to the stock and the number of shares here.
COMPENSATION
In compliance with section 17(b) of the Securities Act we are disclosing that we have been compensated a fee pursuant to an agreement between Smallcaps Daily and IA Media LLC (d/b/a/ “IA Media”) hereinafter referred to as IA Media. Please see IA Media’s disclosure page here. Small Caps Daily was hired by IA Media for a period beginning March 2023 and ending June 2023 to publicly disseminate information about Tiziana Life Sciences Ltd via website, email, and sms. We were paid one hundred seventy-five thousand usd via ACH. Readers are advised to review SEC periodic reports: forms 10Q 10K, form 8K, insider reports, forms 3, 4, 5 schedule 13d. Smallcaps Daily is compliant with the CAN-SPAM Act of 2003. Smallcaps Daily does not offer investment advice or analysis, and Smallcaps Daily further urges you to consult your own independent tax, business, financial, and investment advisors. investing in micro-cap, small-cap, and growth securities is highly speculative and carries an extremely high degree of risk. It is possible that an investor's investment may be lost or impaired due to the speculative nature of the companies profiled. The private securities litigation reform act of 1995 provides investors a safe harbor in regard to forward-looking statements. Any statements that express or involve discussions with respect to predictions, expectations, beliefs, plans, projections, objectives, goals, assumptions or future events, or performance are not statements of historical fact but may be forward-looking statements. Forward-looking statements are based on expectations, estimates, and projections at the time the statements are made that involve a number of risks and uncertainties which could cause actual results or events to differ materially from those presently anticipated. Forward-looking statements in this action may be identified through the use of words such as projects, foresee, expects, will, anticipates, estimates, believes, understands, or that by statements indicating certain actions & quotes; may, could, or might occur. Understand there is no guarantee past performance will be indicative of future results in preparing this publication. Smallcaps Daily has relied upon information supplied by its clients, as well as its clients’ publicly available information and press releases which it believes to be reliable; however, such reliability can not be guaranteed. Investors should not rely on the information contained on this website. Rather, investors should use the information contained in this website as a starting point for doing additional independent research on the featured companies. The advertisements in this website are believed to be reliable, however, Smallcaps Daily and its owners, affiliates, subsidiaries, officers, directors, representatives, and agents disclaim any liability as to the completeness or accuracy of the information contained in any advertisement and for any omissions of material facts from such advertisement. Smallcaps Daily is not responsible for any claims made by the companies advertised herein, nor is Smallcaps Daily responsible for any other promotional firm, its program, or its structure. Smallcaps Daily is not affiliated with any exchange, electronic quotation system, the Securities Exchange Commission, or FINRA.
Copyright © 2023 Smallcaps Daily. All rights reserved.